Conclusions
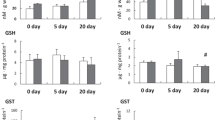
These data show that of the three major constituents characteristic of most hard waters (i.e., Ca2+, Mg2+, and the CO2 system) calcium ion, introduced either as sulfate or carbonate, is the major source of protection against cadmium toxicity. This protective effect is observed in the absence of significant cadmium precipitation. Magnesium ion, sulfate ion, sodium ion and the carbonate system provide little or no protection.
Similar content being viewed by others
References
BROWN, V.M.: Water Res.2, 723 (1968).
HARRIS, E.K.: Biometrics15, 424 (1959).
HIGUCHI, K.: Nichisai Igaku Lasshi33, 899 (1974).
JONES, J.R.E.: J. Exp. Biol.15, 391, (1938).
JONES, J.R.E.: J. Exp. Biol.16, 313 (1939).
KINKADE, M.L. and H.E. EREMAN: Environ. Res.10, 308 (1975).
McCARTHY, L.S., J.A.C. HENRY and A.H. HOUSTON: J. Fish Res. Board Can.35, 35 (1978).
PICKERING, Q.H. and C. HENDERSON: Air Water Pollut. Int. J.10, 453 (1966).
TABATA, K.: Bull. Tokai Region Fisheries Research Lab.58, 215 (1969)
U.S. ENVIRONMENTAL PROTECTION AGENCY: Ecol. Res. Series EPA-660-75-009 (1975).
U.S. ENVIRONMENTAL PROTECTION AGENCY: Methods for Chemical Analysis of Water and Wastes. EPA-625/6-74-003a (1976).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carroll, J.J., Ellis, S.J. & Oliver, W.S. Influences of hardness constituents on the acute toxicity of cadmium to brook trout (Salvelinus fontinalis). Bull. Environ. Contam. Toxicol. 22, 575–581 (1979). https://doi.org/10.1007/BF02026990
Issue Date:
DOI: https://doi.org/10.1007/BF02026990