Abstract
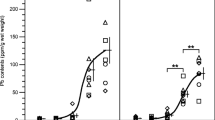
Previous human and experimental studies have demonstrated that lead exposure may modify the metabolism of lipids. Several studies have indicated that exposure to lead produces an increase in lipid peroxidation and inhibits blood superoxide dismutase activity. Recently, lipid peroxides have been shown to impair tissue membranes and to be a risk factor for vascular diseases. The aim of the present investigation was to evaluate the impact of subclinical lead poisoning on rat lipids in the context of atherosclerosis. The degree of poisoning was analogous to that in populations exposed to lead in a contaminated environment. Experiments were performed on male Buffalo rats with body weights of 150–200 g. The experimental animals received lead acetate intragastrically in doses of 35 mg lead/kg body wt. (Pb/kg) once weekly or 70 mg Pb/kg twice weekly for 7 weeks. Control rats were fed in the same manner with sodium acetate equimolar to the acetate in the lead acetate solution. One day after the feeding was over, venous blood samples, under ether anesthesia, were collected. The animals were killed by exsanguination and the liver was excised for determination of the metal (lead, copper, and zinc) content. A segment of the abdominal aorta was excised for histological examination. In venous blood the following were estimated: triglycerides, total cholesterol, high-density lipoprotein (HDL)-cholesterol fraction, serum lipid peroxides, and blood superoxide dismutase activity. Metal content (lead, copper, and zinc) in blood and liver was determined by means of atomic absorption spectrophotometry. In rats poisoned with small doses of lead, decreases in the plasma cholesterol level and the HDL-cholesterol fraction were observed. In parallel with the decrease in the cholesterol concentration, lead increases the serum triglyceride level, this increase being dependent upon lead levels in blood. In our studies a significant influence of lead on serum lipid peroxide level or blood superoxide dismutase activity was not found. In the histological examination, atrophy of the elastic fibers in the aorta was observed. The possible significance of the inhibitory effect of lead on lipoprotein lipase activity is discussed.
Similar content being viewed by others
References
Chajek-Shaul T, Friedman G, Stein O, Shiloni E, Etienne J, Stein Y (1989) Mechanism of the hypertriglyceridemia induced by tumor necrosis factor administration to rats. Biochim Biophys Acta 1001: 316–324
Chung BH, Segrest JP, Smith K, Griffin FM, Brouillette CG (1989) Lipolytic surface remnants of triglyceride-rich lipoproteins are cytotoxic to macrophages but not in the presence of high density lipoprotein. A possible mechanism of atherogenesis? J Clin Invest 83: 1363–1374
Cocco PL, Cocco E, Anni MS, Flore C, Melis A, Salis S (1991) Occupational exposure to lead and blood cholesterol in glucose-6-phosphate dehydrogenase deficient and normal subjects. Res Commun Chem Pathol Pharmacol 72: 81–95
Eckel RH (1989) Lipoprotein lipase. A multifunctional enzyme relevant to common metabolic diseases. N Engl J Med 320: 1060–1068
Gagne C, Brun LD, Ulien P, Moorjani S, Lupien PJ (1989) Primary lipoprotein lipase activity deficiency: clinical investigation of a French Canadian population. Can Med Assoc J 140: 405–411
Hassan AS (1987) The effect of dimethyl sulfoxide on cholesterol and bile acid metabolism in rats. Proc Soc Exp Biol Med 186: 205–210
Ito Y, Niiya I, Kurita H, Shima S, Sarai S (1985) Serum lipid peroxide level and blood Superoxide dismutase activity in workers with occupational exposure to lead. Int Arch Occup Environ Health 56: 119–127
Kihara S, Kubo M, Ikeda N, Yokoyama S, Matsuzawa Y, Tarui S, Yamamoto A, Hostetler KY (1989) Inhibition of purified human postheparin lipoprotein lipase by beta-adrenergic blockers in vitro. Biochem Pharmacol 38: 407–411
Kopp SJ, Barron JT, Tow JP (1988) Cardiovascular action of lead and relationship to hypertension: a review. Environ Health Perspect 78: 91–99
McCord JM, Fridovich J (1969) An enzymic function for erythrocuprein (hemocuprein). J Biol Chem 214: 6049–6055
Qinlan GJ, Halliwell B, Moorhouse CP, Gutteridge JM (1988) Action of lead (II) and aluminium (III) ions on iron stimulated lipid peroxidation in liposomes, erythrocytes and rat liver microsomal fractions. Biochim Biophys Acta 962: 196–200
Satoh K (1978) Serum lipid peroxide in cerebrovascular disorders determined by a new colorimetric method. Clin Chim Acta 90: 37–43
Selander S, Cramer K (1968) Determination of lead in blood by atomic spectrophotometry. Br J Ind Med 25: 209–213
Skoczyńska A, Juzwa W, Smolik R, Szechiński J, Behal F (1986) Response of the cardiovascular system to catecholamines in rats given small doses of lead. Toxicology 39: 275–289
Tandon SK, Flora S (1989) Dose and time effects of combined exposure to lead and ethanol on lead body burden and some neuronal, hepatic and haematopoietic biochemical indices in the rat. J Appl Toxicol 9: 347–352
Wojtczak-Jaroszowa J, Kubow S (1989) Carbon monoxide, carbon disulfide, lead and cadmium — four examples of occupational toxic agents linked to cardiovascular disease. Med Hypotheses 30: 141–150
Xiao GH, Wu JL, Liu YG (1989) The effects of cadmium, mercury and lead in vitro on hepatic microsomal mixed function oxidase and lipid peroxidation. J Tongji Med Univ 9: 81–85
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Skoczyńska, A., Smolik, R. & Jeleń, M. Lipid abnormalities in rats given small doses of lead. Arch Toxicol 67, 200–204 (1993). https://doi.org/10.1007/BF01973308
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01973308