Abstract
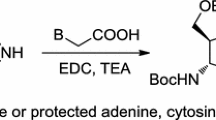
Contrary to reports in the early literature, 5-aminoimidazoles can be prepared in good yield as crystalline compounds. A study of their chemistry has shown that they behave as either C- or N-nucleophiles depending upon the nature of the electrophile. These addition and addition-elimination reactions provide useful new routes to nitrogen heterocycles. An important naturally occurring 5-aminoimidazole, aminoimidazole ribonucleotide (AIR) is a biosynthetic precursor of purines and thiamine. The chemistry of 5-aminoimidazoles is discussed and a synthesis of the aminotmidazole ribonucleoside (AIRS) and its biomimetic transformation to novel purine analogues is presented.
Similar content being viewed by others
References
D. J. Lythgoe and C. A. Ramsden, Adv. Heterocycl. Chem.,61, 1 (1994).
E. J. Mueller, E. Meyer, J. Rudolph, V. J. Davisson, and J. Stubbe, Biochemistry,33, 2269 (1994).
N. J. Cusack, G. Shaw, and G. J. Litchfield, J. Chem. Soc. C, No. 8, 1501 (1971).
V. V. Alevin, T. R. Kostikova, and V. D. Domkin, Zh. Obshch. Khim.,57, 692 (1987).
B. Estramareix and M. Therisod, J. Am. Chem. Soc.,106, 3857 (1984).
G. E. Adams, A. Breccia, and B. Cavalleri (eds.), Nitroimidazoles: Chemistry, Pharmacology and Clinical Application, NATO Advanced Study Institutes Series, Series A: Life Sciences, Plenum Press, New York-London (1982), Vol. 42.
D. I. Edwards, Comprehensive Medicinal Chemistry, Vol. 2, P. G. Sommes (ed.), Pergamon Press, Oxford (1990), pp, 734–742.
W. J. Ehlhardt, B. B. Beaulieu, Jr., and P. Goldman, Biochem. Pharmacol.,36, 259 (1987).
C. E. Sullivan, F. P. Tally, B. R. Goldin, and P. Vouros, Biochem. Pharmacol.,31, 2689 (1982).
J. H. Buyer, Nitroazoles, VCH Publishers Inc., Florida (1986).
A. H. M. Al-Shaar, D. W. Gilmour, D. J. Lythgoe, I. McClenaghan, and C. A. Ramsden, J. Chem. Soc., Perkin Trans, 1, No. 21, 2779 (1992),
R. H. Jones, A. P. Lothian, and C. A. Ramsden (1995), submitted for publication.
A. H. M. Al-Shaar, R. K. Chambers, D. W. Gilmour, D. J. Lythgoe, I. McClenaghan, and C. A. Ramsden, J. Chem. Soc., Perkin Trans. 1,21, 2789 (1992).
R. Huisgen, K. Herbig, A. Siegl, and H. Huber, Chem. Ber.,99, 2526 (1966).
A. D. Broom, J. L. Shim, and G. L. Anderson, J. Org. Chem.,41, 1095 (1976); T. Itoh, I. Fujii, Y. Tomii, H, Nishimura, H. Ogura, and Y, Mizuno, Heterocycles,24, 927 (1986).
B. Bhat, M. P. Groziak, and N. J. Leonard, J. Am, Chem. Soc.,112, 4891 (1990).
C. Chavis, F. Grodenic, and J.-L. Imbach, Eur. J. Med. Chem.,14, 123 (1979).
M. J. Humphries and C. A. Ramsden, Synlett.,2, 203 (1995).
W. J. Humphries and C. A. Ramsden, unpublished results.
Additional information
Department of Chemistry, Keele University, Keele, Staffordshire, ST5 5BG, England. Published in Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1323–1331, October, 1995. Original article submitted July 15, 1995.
Rights and permissions
About this article
Cite this article
Ramsden, C.A. 5-Aminoimidazole chemistry. Chem Heterocycl Compd 31, 1155–1162 (1995). https://doi.org/10.1007/BF01185582
Issue Date:
DOI: https://doi.org/10.1007/BF01185582