Abstract
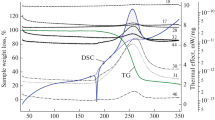
The Arrhenius parameters of the thermal composition of pyridine N-oxides in the liquid phase have been determined. The conclusion has been reached that the formation of an oxaziridine activated complex can be considered as the initial act of the thermolysis. A correlation has been found to exist between the rate constants of the thermal decomposition and the calculated values of the fractional electron charge at the oxygen of the N-oxide, and the length of the N- O bonds.
Similar content being viewed by others
References
I. Katada, Pharm. Soc. Jpn.,67, 53 (1947).
Y. Itoh, A Ohsawa, and H. Igeta, Heterocycles, No. 1, 256 (1986).
T. Itoh, A. Ohsawa, Y. Itoh, and H. Igeta, Heterocycles, No. 5, 783 (1990).
M. J. S. Dewar and W. Thiel, J. Am. Chem. Soc., No. 15, 4899 (1977).
L. Shaofeng and G. Pilcher, J. Chem. Term., No. 4, 463 (1988).
L. Bellamy, New Data on IR Spectra of Complex Molecules [Russian translation], Mir, Moscow (1971), p. 42.
A. I. Gol'binder, Laboratory Experiments for a Course in the Theory of Explosive Substances [in Russian], Rosvuzizdat, Moscow (1963), p. 9.
Additional information
Kazan' State Technological University, Kazan' 420015. Biisk Liceum of the Altai Region, Biisk 659322. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 644–646, May, 1994. Original article submitted April 4, 1994.
Rights and permissions
About this article
Cite this article
Klyuchnikov, O.R., Saifullin, I.S. & Nikishev, Y.Y. Structural kinetic rules governing the thermal decomposition of pyridine N-oxides. Chem Heterocycl Compd 30, 564–566 (1994). https://doi.org/10.1007/BF01169835
Issue Date:
DOI: https://doi.org/10.1007/BF01169835