Abstract
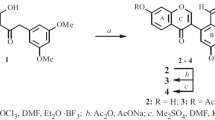
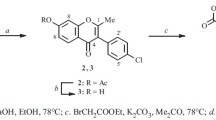
Syntheses are reported far sterically hindered 2-substituted benzodioxane analogs of isoflavone. An optically active lanthanide shift reagent (LSR) was used to show that these products exist as mixtures of atropoisomers. The difference in the chemical shifts of the enantiomeric protons in the presence of the LSR may reach 2–3 ppm with decreasing temperature. Conclusions were drawn concerning the structure of the LSR-substrate adducts and their conformations.
Similar content being viewed by others
References
N. V. Gorbulenko and V. P. Khilya, Ukr. Khim. Zh.,60, No. 1, 709 (1994).
A. L. Kazakov, V. P. Khilya, V. V. Mezheritskii, and D. Litkei, Natural and Modified Isoflavonoids [in Russian], Izd. Rostovsk. Univ., Rostov-on-the-Don (1985).
L. Carollini and G. Lucchetti, Gazz. Med. Ital.,135, Nos. 7-8, 365 (1976).
A. V. Turov, I. V. Komarov, V. P. Khilya, and M. Yu. Kornilov, Teor. Éksperim. Khim., No. 5, 629 (1989).
A. V. Turov and V. P. Khilya, Khim. Geterotsikl. Soedin., No. 6, 723 (1996).
B. Bleany, J. Magn. Reson.,8, 91 (1972).
M. Yu. Kornilov, A. V. Turov, and V. I. Zamkovoi, Ukr. Khim. Zh.,41, No. 7, 769 (1975).
H. M. McConnel and R. E. Robertson, J. Chem. Phys.,29, No. 12, 1361 (1958).
M. Yu. Kornilov, V. V. Plakhotnik, A. V. Turov, and V. P. Khilya, Ukr. Khim. Zh.,58, No. 11, 1026 (1992).
Additional information
Taras Shevchenko Kiev University, 252033 Kiev. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 902–908, July, 1996. Original article submitted March 27, 1996.
Rights and permissions
About this article
Cite this article
Bondarenko, S.P., Turov, A.V. & Khilya, V.P. Synthesis of sterically hindered benzodioxane analogs of isoflavone and a study of their atropoisomerism. Chem Heterocycl Compd 32, 767–772 (1996). https://doi.org/10.1007/BF01165717
Issue Date:
DOI: https://doi.org/10.1007/BF01165717