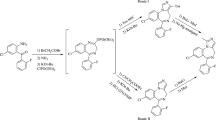
Abstract
The alkylation of 1-aryldihydro-4(IH,3H)-pyrimidinone-2-thiones goes through both at the thiol group and at the amide nitrogen atom of the heterocycle. Enlargement of the alkyl radical favors an increase in the portion of the N-alkyl derivative in the mixture. The influence of the folding of the heterocycle on its barrier to rotation around the Ph-N1 bond was shown.
Similar content being viewed by others
References
V. Yu. Mitskyavichyus, R. S. Baltrushis, I. Ch. Bilinskaite, É. É. Liepin'sh, and R. M. Zolotoyabko, Khim. Geterotsikl. Soedin., No. 9, 1240 (1191).2
R. S. Baltrushis, V. Yu. Mitskyavichyus, I. Ch. Bilinskaite, R. M. Zolotoyabko, and É. É. Liepin'sh, Khim. Geterotsikl. Soedin., No. 8, 1096 (1990).
J. B. Lambert, Acc. Chem. Res., No. 4, 389 (1971).
Additional information
Kaunas Technological University, Kaunas LT-3006. Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1255–1260, September, 1996. Original article submitted March 11, 1996.
Rights and permissions
About this article
Cite this article
Mitskyavichyus, V.Y., Bilinskaite, I.C. Alkyl derivatives of 1-aryldihydro-4(IH,3H)-pyrimidinone-2-thiones and their structure. Chem Heterocycl Compd 32, 1078–1083 (1996). https://doi.org/10.1007/BF01164716
Issue Date:
DOI: https://doi.org/10.1007/BF01164716