Abstract
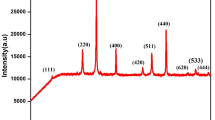
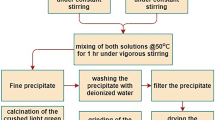
Single phase γ′-Fe4N type (Fe1−xNix)4N compounds (0 ≤ x ≤ 0.6 have been synthesized for the first time by controlled heat treatment of iron-nickel oxalates in a gaseous flow of NH3 xand H2. The preparation processes were investigated using differential scanning calorimetry (DSC), X-ray diffraction and Mössbauer spectroscopy. The results confirmed that annealing of oxalates in the NH3 and H2 atmosphere included the processes of dehydration, decomposition and reduction, nitrogenation and thermal decomposition of the nitrides. The decomposition and reduction occur simultaneously. The final products depend on the flow rate ratio of NH3 ∶ H2 and the annealing temperature. The formation conditions for the single phase γ′-Fe4N type (Fe1−xNix)4N compounds are related to the nickel concentration, with increasing nickel content, the nitrogenation temperature decreased, in contrast the flow rate ratio of NH3∶ H2 increased.
Similar content being viewed by others
References
J. C. Wood, Jr andA. J. Nozik,Phys. Rev. B 4 (1971) 2224.
K. Umeda, E. Kita andA. Tasaki,IEEE Trans. Magn. 22 (1986) 591.
S. Jin, S. K. Hwang andJ. W. Morris, Jr,Metall. Trans. A 6 (1975) 1569.
S. K. Chen, S. Jin, T. H. Tiefel, Y. F. Hsieh, E. M. Gyorgy andD. W. Johnson, Jr,J. Appl. Phys. 70 (1991) 6247.
B. Siberchicot, S. F. Matar, L. Fournés, G. Demazeau andP. Hagenmüller,J. Solid State Chem. 84 (1990) 10.
D. Andriamandroso, L. Fefilative, G. Demazeau, L. Fournes andM. Pouchard,Mater. Res. Bull 19 (1984) 1187.
G. Shirane, W. J. Takei andS. L. Ruby,Phys. Rev. 126 (1962) 49.
P. Mohn, K. Schwarz, S. Mater andG. Demazeau,Phys. Rev. B. 48 (1992) 4000.
S. F. Matar, J. G. M. Armitage, P. C. Riedi, G. Demazeau andP. Hagenmüller,Eur. J. Solid State Inorg. Chem. 26 (1989) 517.
W. J. Schuele,J. Phys. Chem. 63 (1959) 83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, F., Yang, J., Xue, D. et al. Preparation of the single phase γ′-(Fe1−xNix)4N compounds (0 ≤x ≤ 0.6). J Mater Sci 30, 4857–4860 (1995). https://doi.org/10.1007/BF01154495
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01154495