Summary
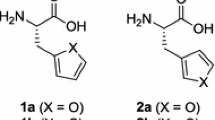
Non-proteinogenic amino acids play an increasing role in oligopeptide chemistry. Their pharmacological and chemical properties, caused by D-configuration and “unnatural” residues, are more and more used for drug design. Different methods of asymmetric synthesis have been developed during the last decade to prepare “unusual” amino acids. One of them, the asymmetric hydrogenation of dehydroamino aids catalyzed by chiral rhodium (I) complexes, will be described. A series of examples, D- and L-configured, like naphthyl-, thienyl-, furyl-, and pyridylalanines, as well as phenylalanines substituted by chlorine, fluorine, p-nitro, p-methyl, p-trifluoromethyl, p-isopropyl, and p-tert-butyl have been prepared and characterized. Some analytical data like melting points and values of optical rotation are summarized in tables.
Similar content being viewed by others
Abbreviations
- (−)-DIOP:
-
(4R,5R)-4,5-Bis(diphenylphosphinomethyl)-2,2-dimethyl-1,3-dioxolane
- (−)-BPPM:
-
(2S,4S)-N-tert-Butoxycarbonyl-4-diphenylphosphino-2-diphenylphosphinomethyl-pyrrolidine
- Ph-β-glup:
-
Phenyl 4,6-O-(R)-benzylidene-2,3-O-bis(diphenylphosphino)-β-D-glucopyranoside
- DuPHOS:
-
1,2-bis-(phospholano)benzene
- PROPRAPHOS:
-
2,3-O,N-bis(diphenylphosphino)-1-(naphthoxy)-2-hydroxy-3-isopropylamino propane
- PINDOPHOS:
-
2,3-O,N-bis(diphenylphosphino)-1-(4-indolyloxy)-2-hydroxy-3-isopropylamino propane
References
Bozell JJ, Vogt CE, Gozum JJ (1991) Transition-metal-assisted asymmetric synthesis of amino acid analogues. A new synthesis of optically pure D- and L-pyridylalanines. J Org Chem 56: 2584–2587
Brunner H, Zettlmeier W (1993) Handbook of enantioselective catalysis. VCH Verlagsgesellschaft, Weinheim
Burk MJ, Feaster JE, Nugent WA, Harlow RL (1993) Preparation and use of C2-symmetric bis(phospholanes): production ofα-amino acid derivatives via highly enantioselective hydrogenation reactions. J Am Chem Soc 115: 10125–10138
Burk JM, Gross MF, Martinez JP (1995) Asymmetric catalytic synthesis of β-branched amino acids via highly enantioselective hydrogenation reactions. J Am Chem Soc 117: 9375–9376
Cativiela C, Fernandez J, Mayoral JA, Melendez E, Uson R, Oro LA, Fernandez MJ (1982) Asymmetric hydrogenation of 2-benzamido(acetamido)-3-(2-thienyl)-2-propenoic acids catalyzed by rhodium-DIOP complexes. J Mol Catal 16: 19–25
Cativiela C, Diaz de Villegas MD, Mayoral JA, Melendez E (1983) Synthesis and stereospezific ring opening of the (Z)-(E)-isomers of 2-methyl(phenyl)-4-thienylmethylene)5(4H)-oxazolones. Synthesis: 899–902
Cativiela C, Mayoral JA, Melendez E, Oro LA, Pinillos MT, Uson RJ (1984) Influence of the heterocyclic ring on the asymmetric synthesis of β-hetarylalanines by homogeneous catalytic hydrogenation. J Org Chem 49: 2502–2504
Cativiela C, Diaz de Villegas MD, Garcia JI, Mayoral JA, Melendez E (1985) Sintesis estereoselectiva de acidos (Z)-2-acilamino-3-hetaril-2-propenoicos. An Quim 81: 56–61
Cesarotti E, Chiesa A, Ciani G, Siroui A (1983) Asymmetric hydrogenation catalyzed by aminophosphine-phosphinite-rhodium complexes derived from natural aminoalcohols and X-ray crystal structure of (1,5-cyclooctadiene)-(S)-N-(diphenylphosphino)-2-diphenylphosphinoxymethylpyrrolidinerhodium(I) perchlorate. J Organomet Chem 251: 79–91
Corey EJ, Link O (1992) A general, catalytic, and enantioselective synthesis ofα-amino acids. J Am Chem Soc 114: 1906–1908
Crawford M, Little WT (1959) The Erlenmeyer reaction with aliphatic aldehydes, 2-phenyloxazol-5-one being used instead of hippuric acid. J Chem Soc: 729–731
Döbler Chr, Kreuzfeld HJ, Pracejus H (1988) Chiral bicyclic O,N-bis (diphenylphosphino)-aminoalkanols as ligands for enantioselective metal complex hydrogenation catalysts. J Organomet Chem 344: 89–92
Döbler Chr, Kreuzfeld HJ, Krause HW, Michalik M (1993) Unusual amino acids IV. Asymmetric synthesis of thienylalanines. Tetrahedron: Asymmetry 4: 1833–1842
Döbler Chr, Kreuzfeld HJ, Michalik M, Krause HW (1996) Unusual amino acids VII. Asymmetric synthesis of 3- and 4-pyridylalanines. Tetrahedron: Asymmetry 7: 117–125
Fryzuk MD, Bosnich B (1977) Asymmetric synthesis. Production of optically active amino acids by catalytic hydrogenation. J Am Chem Soc 99: 6262–6267
Halpern J (1985) Asymmetric catalytic hydrogenation: mechanism and origin of enantioselection. Asymmetric synthesis, vol 5. In: Morrison JD (ed) Academic Press, New York, pp 41–68
Hellmann H, Piechota H (1960) Die Aldolreaktion aromatischer und heterocyclischer Aldehyde mit Acetaminomalonsäure-monoestern. Liebigs Ann Chem 631: 175–179
Hengartner U, Valentine Jr D, Johnson KK, Larscheid ME, Pigott F, Scheidl F, Scott JW, Sun RS, Townsend JM, Williams TH (1979) Synthesis of (R)-6-methyltryptophan via enantioselective catalytic hydrogenation. J Org Chem 44: 3741–3747
Hruby VJ, Al-Obeidi F, Kazmierski W (1990) Emerging approaches in the molecular design of receptor-selective peptide ligands: conformational, topographical and dynamic considerations. Biochem J 268: 249–262
Hunt S (1992) Non-protein amino acids. In: Rogers LJ (ed) Amino acids, proteins and nucleic acids, ch. 1. Academic Press, London New York, pp 1–52 (In: Dey PM, Harborne JB (eds) Methods in plant biochemistry, vol 5)
Kagan HB, Dang TP (1972) Asymmetric catalytic reduction with Transition metal complexes. I. A catalytic system of rhodium(I) with (−)-2,3-O-isopropylidene-2,3-dihydroxy-1,4-bis(diphenylphosphino)butane, a new chiral diphosphine. J Am Chem Soc 94: 6429–6433
Karim A, Mortreux A, Petit F (1986) Hydrogenation asymmetrique des deshydroaminoacides catalyses par des complexes rhodium-aminophosphine phosphinite. Influence de la structure des chelates. J Organomet Chem 312: 375–381
Koenig MD (1985) The applicability of asymmetric homogeneous catalytic hydrogenation in asymmetric synthesis, vol 5, ch 3. In: Morrison JD (ed) Academic Press, New York, pp 71–98
Krause HW, Foken H, Pracejus H (1989) Aminophosphine phosphinites of propranolol as ligands for rhodium catalyzed asymmetric hydrogenation of dehydroamino acids. New J Chem 13: 615–620
Krause HW, Kreuzfeld HJ, Döbler Chr (1992a) Asymmetric synthesis of fluorine containing phenylalanines. Tetrahedron: Asymmetry 3: 555–566
Krause HW, Wilcke FW, Kreuzfeld HJ, Döbler Chr (1992b) Unusual amino acids I. Asymmetric synthesis of furylalanine derivatives. Chirality 4: 110–115
Krause HW, Kreuzfeld HJ, Schmidt U, Döbler Chr, Michalik M, Taudien S, Fischer Chr (1996) Unusual amino acids VI. Substituted arylamino acids by asymmetric hydrogenation of N-Cbz and N-Boc protected dehydroamino acid derivatives. Chirality 8: 173–188
Kreuzfeld HJ, Döbler Chr, Krause HW, Facklam Chr (1993) Unusual amino acids V. Asymmetric hydrogenation of (Z)-N-acylamidocinnamic acid derivatives bearing different protective groups. Tetrahedron: Asymmetry 4: 2047–2051
Kreuzfeld HJ, Schmidt U, Döbler Chr, Krause HW (1996) Enantioselective hydrogenation of dehydroamino acid derivatives using PINDOPHOS-rhodium as chiral catalyst. Tetrahedron: Asymmetry 7: 1011–1018
Lubec G, Rosenthal GA (1990) Amino acids, chemistry, biology and medicine. ESCOM, Leiden, pp 3–29
Miyazawa T, Takashima K, Yamada T, Kuwata S, Watanabe H (1982) Studies of unusual amino acids and their peptides. XIV. The asymmetric hydrogenation of the phenylhydrazone of methyl N-(3,3-dimethyl-2-oxobutanoyl)-L-valinate. Bull Chem Soc Jpn 55: 341–342
Morgan BA, Bower JD, Guest KP, Handa BK, Metcald G, Smith CFC (1977) Structure-activity relationship of enkephalin analogs. In: Goodman M, Meienhofer J (eds) Wiley, New York (Pept Proc Am Pept Symp, 5th: 111–113)
Mortreux A, Petit F, Bruno G, Pfeiffer G, Siv C (1986) Synthesis of chiral aminophosphine phosphinites. Use in catalytic asymmetric reduction. J Organomet Chem 317: 93–104
Nagel, U, Kinzel E, Andrade I, Prescher G (1986) Synthese N-substituierter (R,R)-3,4-Bis(diphenylphosphino)-pyrrolidine und Anwendung ihrer Rhodiumkomplexe zur asymmetrischen Hydrierung vonα-(Acylamino)acrylsäure-Derivaten. Chem Ber 119: 3326–3343
Noda K, Shimohigashi Y, Izumiya N (1983)α,β-Dehydroamino acids and peptides. Peptides (N.Y.) 5: 285–339
Noyori R (1994) Asymmetric catalysis in organic synthesis, ch 2. Wiley and Sons, New York, pp 16–94
Pinski J, Schally AV, Yano T, Groot K, Srkalovic G, Serfozo P, Reissmann Th, Bernd M, Deger W, Kutscher B, Engel J (1995) Evaluation of the in vitro and in vivo activity of the L-, D,L- and D-Cit6 forms of the LH-RH antagonist Cetrorelix (SB-75). Int J Peptide Protein Res 45: 410–417
Pracejus G, Pracejus H (1984) Chiral O,N-bis(diphenylphosphino)aminoalkanols as ligands for enantioselective metal complex hydrogenation catalysts. J Mol Catal 24: 227–230
Pracejus H, Pracejus G, Costisella B (1987) O,N-Bis(diphenylphosphino)derivatives of chiral trans- and cis-2-aminocyclohexanols: synthesis and enantioselective behaviour as ligands in Rh-based homogeneous hydrogenation catalysts. J Prakt Chem 329: 235–245
Rao YS, Filler R (1975) Geometric isomers of 2-aryl(aralkyl)-4-arylidene(alkylidene)-5(4H)-oxazolones. Synthesis: 749–764
Reissmann Th, Engel J, Kutscher B, Bernd M, Hilgard P, Peukert M, Szelenyi I, Reichert S, Gonzales-Barcena D, Nieschiag E, Comaru-Schally AM, Schally AV (1994) Cetrorelix (SB-75) LH-RH antagonist. Drugs of the Future 19: 228–237
Schmidt U, Häusler J, Oehler E (1979) Dehydroamino acids. 2-Hydroxy-2-amoino acids and 2-mercapto-2-amino acids. Fortschr Chem Org Naturst 37: 251–327
Schmidt U, Lieberknecht A, Wild J (1988) Didehydroamino acids (DDAA) and didehydropeptides (DDP). Synthesis: 159–172
Schmidt U, Griesser H, Leitenberger V, Lieberknecht A, Mangold R, Meyer R, Riedl B (1992a) Diastereoselective formation of (Z)-didehydroamino acid esters. Synthesis: 487–490
Schmidt U, Meyer R, Leitenberger V, Griesser H, Lieberknecht A (1992b) Amino acids and peptides. 84. Synthesis of biologically active cyclopeptides. 24. Total synthesis of the biphenomycins. III. Synthesis of biphenomycin B. Synthesis: 1025–1030
Seebach D, Dziadulewicz E, Behrendt L, Cantoreggi S, Fitzi R (1989) Synthesis of nonproteinogenic (R)- or (S)-amino acids; analogues of phenylalanine, isotopically labelled and cyclic amino acids from tert-butyl-2-(tert-butyl)-3-methyl-4-oxo-1-imidazolidine carboxylate. Liebigs Ann Chem: 1215–1232
Seebach D, Bürger HM, Schickli CP (1991) Stereoselektive Umsetzungen von rac-, (R)- oder (S)-5-Alkyliden-2-t-butyl-3-methoxy-4-oxo-1-imidazolidincarbonsäure-t-butylestern (chirale 2,3-Dehydroaminosäure-Derivate) und Herstellung einiger nichtproteinogener Aminosäuren. Liebigs Ann Chem: 669–684
Takaya H, Ohta T, Noyori R (1993) Asymmetric hydrogenation, in catalytic asymmetric synthesis, ch. 1 (and ref. therein) In: Ojima I (ed) VCH Verlagsgesellschaft, Weinheim
Taudien S, Schinkowski K, Krause HW (1993) Unusual amino acids III. Asymmetric synthesis of 3-arylalanines. Tetrahedron: Asymmetry 4: 73–84
Wagner I, Musso H (1993) Natürliche Aminosäuren. Angew Chem 95: 827–839
Wenger RM (1986) Cyclosporine and analogs. Isolation and synthesis. Mechanism of action and structural requirements for pharmacological activity. Progress Chem Org Nat Prods 50: 123
Williams RM (1989) Synthesis of optically activeα-amino acids. Pergamon Press, Oxford
Wynants C, Coy DH, van Binst G (1988) Conformational study of super-active analogues of Somatostatin with reduced ring size by1HNMR. Tetrahedron 44: 941–973
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kreuzfeld, H.J., Döbler, C., Schmidt, U. et al. Synthesis of non-proteinogenic (D)- or (L)-amino acids by asymmetric hydrogenation. Amino Acids 11, 269–282 (1996). https://doi.org/10.1007/BF00807936
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807936