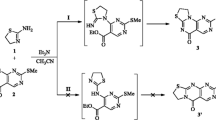
Abstract
Alkaline hydrolysis of substituted 2-cyanocinnamic acids leads to the corresponding substituted 3-carboxymethylpthalimidines; acid hydrolysis of the same acids results in the formation of phthalides with an analogous structure. NMR and mass spectrometric data are examined critically.
Similar content being viewed by others
Literature cited
F. S. Babichev and V. A. Kovtunenko,The Chemistry of Isoindole [in Russian], Naukova Dumka, Kiev (1983).
A. P. Stankyavichyus and A. N. Kost, Nauchn.Tr. Vyssh. Uchebn. Zaved. Lit. SSR, Ser. Khim. Khim. Tekhnol.,23, 163 (1971).
A. P. Stankyavichyus and A. N. Kost,Zh. Org. Khim.,6, 1022 (1970).
G. É. Dudenas, A. P. Stankyavichyus, and A. N. Kost,Zh. Org. Khim.,13, 2185 (1977).
A. P. Stankyavichyus, G. É. Dudenas, and I. I. Shulyakene,Summaries of Papers from Republic Conference on Chemistry and Technology of Organic Materials [in Russian], Kaunas (1981), p. 60.
D. S. Noyce, P. A. King, F. B. Kirby, and W. L. Reed,J. Am. Chem. Soc.,84, 1632 (1962).
R. P. Linstead and F. T. Weiss,J. Chem. Soc., No. 8, 2975 (1950).
E. N. Zil'berman,Reactions of Nitriles [in Russian], Khimiya, Moscow (1972), p. 87.
P. B. Terent'ev,Mass Spectrometry in Organic Chemistry [in Russian], Vysshaya Shkola, Moscow (1979), p. 75.
B. Schaldach and H. Fr. Grutzmacher,Org. Mass. Spectrom.,15, 175 (1980).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 472–476, April, 1992.
Rights and permissions
About this article
Cite this article
Stankyavichyus, A.P., Terent'ev, P.B. & Anyulis, A.S. Synthesis and spectral properties of phthalimidines and phthalides. Chem Heterocycl Compd 28, 396–399 (1992). https://doi.org/10.1007/BF00766995
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00766995