Abstract
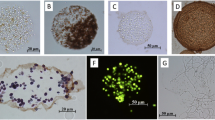
A composite gel system has been developed combining the chemical and physical properties of calcium alginate and agarose gels. The results of growing composite gel immobilized hybridoma SPO1 cells in a protein-free medium within a fluidized-bed perfusion bioreactor are presented in this paper. During the continuous operation of this system, the total cell density reached 3.9×107 cells per ml of beads (viability 79.6%). The specific productivity of monoclonal antibody of the immobilized hybridoma cells reached more than 1.5 μg per 106 viable cells per hour, compared with 0.5 for non-immobilized viable cells grown in a one liter agitated bioreactor with the same medium. Significant increases in cell metabolic activities, including substrate utilization and byproduct formation, were also observed. Leaching of materials from the beads was evident and the major fraction of released materials was alginate.
Similar content being viewed by others
References
Al-Rubeai MA, Musgrave SC, Lambe CA, Walker AG, Evans NH & Spier RE (1990) Methods for the estimation of the number and quality of animal cells immobilized in carbohydrate gels. Enzyme Microbial. Technol. 12: 459–463.
Arnott S, Fulmer A, Scott WE, Dea ICM, Moorhouse R & Rees DA (1974) The agarose double helix and its function in gel structure. J. Mol. Biol. 90: 269–284.
Audet P & Lacroix C (1989) Two-phase dispersion process for the production of biopolymer gel beads: effects of various parameters on the bead size and their distribution. Process Biochem. P. 217–226.
Damsky CM & Bernfield M (1991) Cell-to-cell contact and extracellular matrix. Current Opinion in Cell Biol. 3: 777–778.
Domurado M, Deramoudt F-X & Thomas D (1988) Immobilization of murine lymphocytes by gel entrapment. Biotechnol. Lett. 2(3): 211–216.
Familletti PC & Fredericks JE (1988) Techniques for mammalian cell immobilization. Bio/Technol. 6: 41–44.
Fukushima Y, Okamura K, Imai K & Motai H (1988) A new immobilization technique of whole cells and enzymes with colloidal silica and alginate. Biotechnol. Bioeng. 32: 584–594.
Haug A, Larsen B & Smidsrod O (1974) Uronic acid sequence in alginate from different sources. Carbohydrate Res. 32: 217–225.
Kamihira M, Yoshida H, Lijima S & Kobayashi T (1990) Effects of oxygen aeration on production of monoclonal antibody in immobilized hybridoma cell bioreactor. J. Ferment. Bioeng. 69(5): 311–312.
Knight P (1989) Hollow fiber bioreactors for mammalian cell culture. Bio/Technol. 7: 459–461.
Lee GM & Palsson BO (1990) Immobiliozation can improve the stability of the hybridoma antibody productivity in serum-free media. Biotech. Bioeng. 36: 1049–1055.
Miles BJ & Reading AH (1988) Composite material. Brit. UK pat. GB 2, 201, 966.
Nilsson K, Scheirer W, Merten OW, Östberg L, Liehl E, Katinger HWD & Mosbach K (1983) Entrapment of animal cells for production of monoclonal antibodies and other biomolecules. Nature. 302: 629–630.
Runstadle PW, Ozturk SS & Ray NG (1992) An evaluation of hybridoma cell specific productivity: Perfused immobilized, continuous suspension, and batch suspension cultures. In: Murakami H, Shirahata S & Tachibana H (eds.) Animal Cell Technology: Basic & Applied Aspects. 4: 57–63.
Shen BQ (1991) Immobilisation of hybridoma cells. Ph.D. thesis, The University of Queensland, Brisbane, Australia.
Shen BQ, Reid S & Greenfield PF (1992) Continuous monoclonal antibody production by a composite gel perfusion bioreactor in a protein-free medium. In: Murakami H, Shirahata S & Tachibana H (ed.) Animal Cell Technology: Basic & Applied Aspects. 4: 173–178.
Shen BQ, Reid S & Greenfield PF (1993) Examination of polysaccharides for their uses in bybridoma cell culture. Biotechnol. Lett. 7(3): 193–198.
Shen BQ, Greenfield PF & Reid S. Calcium alginate immobilized hybridomas grown using a fluidized-bed perfusion system with a protein-free medium, Cytotechnology (in press).
Shirai Y, Hashimoto K, Yamaji H & Tokashiki M (1987) Continuous production of monoclonal antibody with immobilized hybridoma cells in an expanded-bed fermenter. Appl. Microbiol. Biotechnol. 26: 495–499.
Sinacore MS, Creswick BC & Buehler R (1989) Entrapment and growth of murine hybridoma cells in calcium alginate gel microbeads. Bio/Technol. 7: 1275–1279.
Tanaka H, Matsumura M & Veliky IA (1984) Diffusion characteristics of substrates in Caalginate gel beads. Biotechnol. Bioeng. 26: 53–58.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Shen, B.Q., Greenfield, P.F. & Reid, S. Hybridoma cells in a protein-free medium within a composite gel perfusion bioreactor. Cytotechnology 16, 51–58 (1994). https://doi.org/10.1007/BF00761779
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00761779