Abstract
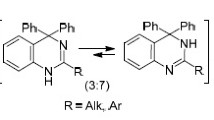
Reaction of 1,2,3,4-tetrahydroisoquinoline enaminoamides with oxalyl chloride gives 2,3-dioxpyrrolo[2,1-a]isoquinolines which react with o-phenylenediamine to give spiro benzimidazolines or condensed quinoxalines, depending on the conditions used.
Similar content being viewed by others
Literature Cited
V. S. Shklyaev, B. B. Aleksandrov, A. G. Mikhailovskii, and M. I. Vakhrin,Khim. Geterotsikl. Soedin., No. 7, 963 (1987).
T. J. Sano,Synt. Org. Chem. Jpn. 42, 340 (1984).
A. V. Ivashchenko, A. G. Drushlyak, and V. V. Tutov,Khim. Geterotsikl. Soedin., No. 5, 667 (1984).
V. S. Shklyaev, B. B. Aleksandrov, M. I. Vakhrin, and G. I. Legotkina,Khim. Geterotsikl. Soedin., No. 11, 1560 (1983).
B. B. Aleksandrov, M. S. Gavrilov, M. I. Vakhrin, and V. S. Shklyaev,Khim. Geterotsikl. Soedin., No. 6, 794 (1985).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 808–810, June, 1990.
Rights and permissions
About this article
Cite this article
Mikhailovskii, A.G., Shklyaev, V.S. & Aleksandrov, B.B. Reaction of 2,3-dioxopyrrolo[2,1-a]isoquinolines with o-phenylenediamine. Chem Heterocycl Compd 26, 674–676 (1990). https://doi.org/10.1007/BF00756422
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00756422