Abstract
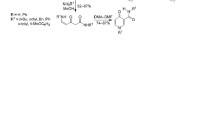
3, 5-Dibromocomanic acid (3, 5-dibromo-γ -pyrone -2-carboxylic acid) VI and its ethyl ester, hitherto not described in the literature, are synthesized by the following route: diethyl acetonedioxalate (I) → diethyl dibromochelidonate (III) → (mono) ethyl dibromochelidonate (IV) → diethyl 3, 5-dibromocomanate (V) → VI. Direct bromination of diethyl bromochelidonate gives the ester III. 6-Bromocomenic acid and its ethyl ester are prepared, as well as 2-bromo-3-hydroxy -γ -pyrone. Bromocomanic acid (x-bromo-γ-pyrone-2-carboxylic acid) XVI is synthesized by the following route: I → (mono) ethyl chelidonate → ethyl comanate → comanic acid XVI. Oxonium salts are obtained: acid sulfates of comanic acid and ethyl comanate.
Similar content being viewed by others
References
G. A. Garkusha, ZhOrKh, 1, 222, 1965.
F. Feist and E. Baum, Ber., 38, 3562, 1905.
A. Thomas and R. Pietrulla, Ber. Pharm. Geselsch., 31, 4, 1921.
J. P. Wiebaut and R. J. C. Kleipool, Rec. trav. chim., 66, 24, 1947.
G. A. Garkusha and G. A. Khutornenko, ZhOKh, 33, 3579, 1963.
G. A. Garkusha and G. A. Khutornenko, ZhOKh, 35, 133, 1965.
L. Ya. Yanovskaya, DAN, 71, 693, 1950.
E. L. Shimmini and F. Challenger, J. Chem., 1185, 1949.
K. Heyns and G. Vogelsang, 87, 1440, 1954.
G. A. Garkusha, ZhOKh, 23, 1578, 1953.
H. Ost, J. Pr. Chem. (2), 23, 440, 1881.
G. A. Garkusha and G. A. Khutornenko, Author's Certificate, 158 273, 1962.
H. Ost, J. Pr. Chem. (2), 29, 62, 1884.
Additional information
For Part VII [1].
Rights and permissions
About this article
Cite this article
Garkusha, G.A., Khutornenko, G.A. γ-Pyrone derivatives. Chem Heterocycl Compd 2, 245–247 (1967). https://doi.org/10.1007/BF00742356
Issue Date:
DOI: https://doi.org/10.1007/BF00742356