Conclusions
1. Several isomeric forms of triglycosides have been found in the natural flavonoid product robinin.
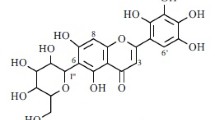
2. The three-stage discontinuous hydrolysis of robinin has given individual fragments in the form of mono-, bi-, and diglycosides showing the structure of the initial isomeric robinin triosides. Six intermediate glycosides have been isolated preparatively: biorobin, diorobin, α-rhamnorobin, β-rhamnorobin, α-rhamnoisorobin, and galactorobin. The structures of the glycosides isolated have been established.
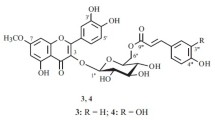
3. An investigation of the intermediate products of the hydrolysis of robinin has shown that robinin contains not less than four triosides—α- and β-neorobinins and α- and β-isorobinins.
4. All the forms of the robinin triosides contain a biose at C-3 differing from the robinobiose described in the literature by the size of the oxide rings of the L-rhamnose and D-galactose. This biose, which we have called neorobinobiose, contains the furanose forms of L-rhamnose and D-galactose and also a β-configuration of the links both between the monoses and between the galactose and the kaempferol.
5. The different forms of the robinins differ from one another by the configurations of the glycosidic links and by the sizes of the oxide rings of the L-rhamnose at C-7.
Similar content being viewed by others
References
N. Wallaschko, Arch. Pharm.,242, 210, 1904.
G. Zemplen and R. Bognar, Berlin,74, 1783, 1941.
N. P. Maksyutina, KhPS [Chemistry of Natural Compounds],1, 63, 1965.
N. P. Maksyutina and V. I. Litvinenko, Dopovidi AS URSR [Ukrainian], 5, 63, 1967.
N. P. Maksyutina, V. I. Litvinenko, and I. P. Kovalev, KhPS [Chemistry of Natural Compounds],2, 388, 1966.
I. P. Kovalev and V. I. Litvinenko, KhPS [Chemistry of Natural Compounds],1, 233, 1965.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 5, No. 4, pp. 227–234, 1969
Rights and permissions
About this article
Cite this article
Maksyutina, N.P. The composition of robinin fromRobinia viscosa . Chem Nat Compd 5, 193–198 (1969). https://doi.org/10.1007/BF00683828
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00683828