Abstract
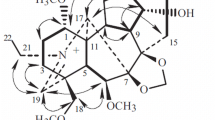
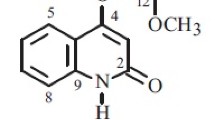
From the combined chloroform-extracted material of the epigeal part ofKorolkowia sewerzowii Regel the new alkaloid sevedamine has been isolated, with mp 256–258°C (acetone) [α]D +3.6° (c 1.25; ethanol), C27H45NO3. On the basis of the results of a study of the UV, IR, PMR, and mass spectra of sevedamine and also performance of a transition to the known alkaloid korseverilinedione it has been established that sevedamine has the structure and configuration of 3β,6α,14α-trihydroxycevanine.
Similar content being viewed by others
Literature cited
K. Samikov, R. Shakirov, and S. Yu. Yunusov, Khim. Prir. Soedin., 367 (1976).
K. Samikov, R. Shakirov, and S. Yu. Yunusov, Khim. Prir. Soedin., 673 (1977).
H. Budzikiewicz, Tetrahedron,20, 2264 (1964).
R. N. Nuriddinov and S. Yu. Yunusov, Khim. Prir. Soedin., 258 (1968).
R. N. Nuriddinov and S. Yu. Yunusov, Khim. Prir. Soedin., 773 (1981).
T. M. Moynehan, K. Schofield, R. A. V. Jones, and A. R. Kaktritsky, J. Chem. Soc., 2637 (1962).
R. N. Nuriddonov, A. I. Saidkhodzhaev, M. R. Yagudaev, and S. Yu. Yunusov, Khim. Prir. Soedin., 333 (1968).
R. N. Nuriddinov and S. Yu. Yunusov, Khim. Prir. Soedin., 333 (1968).
R. F. Zurcher, Helv. Chim. Acta,46, 2054 (1963).
J. Fried, P. Grabovich, R. F. Sabo, and A. I. Cohen, Tetrahedron,20, 2297 (1964).
Additional information
Institute of the Chemistry of Plant Substances of the Uzbek SSR Academy of Sciences, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 622–624, September–October, 1986.
Rights and permissions
About this article
Cite this article
Samikov, K., Shakirov, R. & Yunusov, S.Y. Structure of sevedamine. Chem Nat Compd 22, 585–586 (1986). https://doi.org/10.1007/BF00599268
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00599268