Abstract
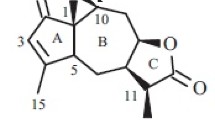
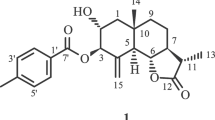
The epigeal part ofErigeron khorassanicus Boiss. has yielded a new sesquiterpene lactone of the pseudoguaiane type — ergolide C17H22O5, mp 179–180°C (ethanol) [α] 20D +123° (c 4.88; ethanol). On the basis of chemical transformations and spectral characteristics, its structure and configuration have been established as 6-acetoxy-4-oxo-1,7αH,6,8,10βH-pseudoguai-11(13)-en-8,12-olide.
Similar content being viewed by others
Literature cited
P. V. Chugunov, V. I. Sheichenko, A. I. Ban'kovskii, and K. S. Rybalko, Khim. Prir. Soedin., 276 (1971).
A. Ovezduryev, B. Akyev, and Sh. V. Kasymov, Khim. Prir. Soedin., 396 (1981).
G. C. Levy and G. L. Nelson, Carbon-13 in Nuclear Magnetic Resonance for Organic Chemists, Wiley-Interscience, New York (1972).
F. Bohlmann, Laxmi, N. Misra, and J. Jakupovic, Phytochemistry,24, No. 5, 1021 (1985).
P. I. Evstratova, K. S. Rybalko, V. I. Sheichenko, and A. I. Ban'kovskii, Khim. Prir. Soedin.,270 (1971).
M. Maruyama and S. Omura, Phytochemistry,16, No. 6, 782 (1977).
P. Kok, P. J. De Clercq, and M. E. Vandewalle, J. Org. Chem.,44, No. 25, 4553 (1979).
K. Nagao, M. Chiba, and S.-W. Kim, Chem. Pharm. Bull.,31, No. 2, 414 (1983).
F. Bohlmann and P. K. Mahanta, Phytochemistry,18, No. 5, 887 (1979).
Additional information
Institute of the Chemistry of Plant Substances of the Uzbek Academy of Sciences, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 568–571, September–October, 1986.
Rights and permissions
About this article
Cite this article
Ovezdurdyev, A., Abdullaev, N.D., Kasymov, S.Z. et al. Structure of ergolide. Chem Nat Compd 22, 532–535 (1986). https://doi.org/10.1007/BF00599255
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00599255