Abstract
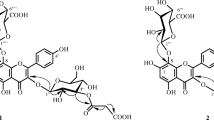
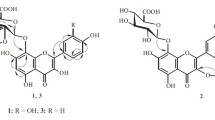
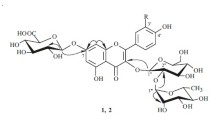
The structure of four new flavonol glycosides isolated from the epigeal part ofRhodiola rosea have been established: 7-O-α-L-rhamnopyranosylgossypetin (rhodiolgin), 8-O-β-D-glucopyranosyl-7-O-α-L-rhamnopyranosylgossypetin (rhodiolgidin), 8-O-β-D-glucopyranosol-7-O-α-L-rhamnopyranosylherbacetin (rhodionidin), and 3-O-β-D-glucopyranosyl-8-O-β-D-xylopyranosylherbacetin (rhodalidin). The properties of the previously undescribed incomplete methyl ethers of herbacetin and gossypetin obtained during the structural analysis of the glycosides have been studied. It has been found that diazomethane methylates the 5-OH groups in the diglycosides investigated.
Similar content being viewed by others
Literature cited
V. A. Kurkin, G. G. Zapesochnaya, and A. N. Shchavlinskii, Khim. Prir. Soedin., 657 (1984).
G. G. Zapesochnaya and V. A. Kurkin, Khim. Prir. Soedin., 23 (1983).
J. H. Bowie and D. W. Cameron, Aust. J. Chem.,19, 1627 (1966).
D. G. I. Kingston, Tetrahedron,27, 2691 (1971).
J. H. Bowie and R. Y. White, J. Chem. Soc. (B), 89 (1969).
R. D. Schmid, R. Mues, et al., Phytochemistry,12, 2765 (1973).
G. P. Kononenko, S. A. Popravko, et al., Bioorg. Khim.,6, 267 (1980).
T. J. Mabry, K. R. Markham, and M. B. Thomas, The Systematic Identification of Flavonoids, Springer, Berlin (1970).
S. Tandon and R. P. Rastogi, Phytochemistry,16, 1455 (1977).
R. M. Dawson, C. A. Hendrick, et al., Aust. J. Chem.,18, 1871 (1965).
T. T. Pangarova and G. G. Zapesochnaya, Khim. Prir. Soedin., 712 (1975).
J. B. Harborne, T. J. Mabry, and H. Mabry, The Flavonoids, Chapman and Hall, London (1975).
V. I. Bykov, V. I. Glyzin, and A. I. Ban'kovskii, Khim. Prir. Soedin., 715 (1972).
Z. Jerzmanowska and J. Kamecki, Roczn. Chem. Ann. Soc. Chim. Pol.,47, 1629 (1973).
P. K. Jauhari, S. C. Sharma, et al., Phytochemistry,18, 359 (1979).
H. Geiger, S. Reichert, and K. R. Markhan, Z. Naturforsch.,37b, 504 (1982).
T.-S. Wu and H. Furukawa, Phytochemistry,22, 1061 (1983).
G. G. Zapesochnaya, Khim. Prir. Soedin., 695 (1982),
G. G. Zapesochnaya, Khim. Prir. Soedin., 21 (1979).
G. G. Zapesochnaya, Khim. Prir. Soedin., 519 (1978).
F. Wessely and G. H. Moser, Monatsh. Chem.,56, 97 (1930).
G. G. Zapesochnaya and T. T. Pangarova, Khim. Prir. Soedin. 554 (1973).
J. B. Harborne and H. T. Clifford, Phytochemistry,8, 2071 (1969).
K. R. Markham and L. J. Porter, Phytochemistry,14, 1093 (1975).
K. R. Markham and L. J. Porter, Pytochemistry,18, 611 (1979).
V. M. Chari, R. J. Grayer-Barkmeijer, et al., Phytochemistry,20, 1977 (1981).
T. A. Geissman, The Chemistry of Flavonoid Compounds, Pergamon, Oxford, (1962), p. 184.
G. P. Shnyakina and G. G. Zapesochnaya, Khim. Prir. Soedin., 92 (1975).
J. B. Harborne, Phytochemistry,8, 177 (1969).
Additional information
All-Union Scientific-Research Institute of Medicinal Plants, Moscow. Translated from Khimiya Prirodnykh Soedinenii. No. 4, pp. 496–507, July–August, 1985.
Rights and permissions
About this article
Cite this article
Zapesochnaya, G.G., Kurkin, V.A. & Shchavlinskii, A.N. Flavonoids of the epigeal part ofRhodiola rosea. II. Structures of new glycosides of herbacetin and of gossypetin. Chem Nat Compd 21, 464–473 (1985). https://doi.org/10.1007/BF00579139
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00579139