Abstract
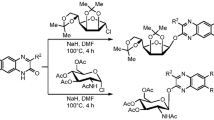
N4-Benzoylcytosine 1-(methyl 2,3,4-tri-O-acetyl-β-D-glucopyranosiduronate) has been obtained with a yield of 70% by the glycosylation of the trimethylsilyl derivative of N4-benzoylcytosine with methyl 1,2,3,4-tetra-O-acetyl-β-D-glucopyranuronate in the presence of three equivalents of SnCl4 as condensing agent. Cytosine 1-(β-D-glucopyranosiduronamide) (IV) — the amide of pentopyranic acid — has been synthesized in practically quantitative yield by the ammonolysis of the nucleoside (I).
Similar content being viewed by others
Literature cited
H. Seto, K. Furihata, and H. Yonehara, J. Antibiot.,29, 595 (1976).
K. A. Watanabe, D. H. Hollenberg, and J. J. Fox, J. Antibiot.,29, 597 (1976).
P. C. Wyss, J. Kiss, and W. Arnold, Helv. Chim. Acta,58, No. 6, 1847 (1976).
Additional information
Peat Institute, Academy of Sciences of the Belorussian SSR, Minsk. Institute of Bioorganic Chemistry, Academy of Sciences of the Belorussian SSR, Minsk. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 617–619, September–October, 1983.
Rights and permissions
About this article
Cite this article
Kulinkovich, L.N., Timoshchuk, V.A. Synthesis of derivatives of pentopyranic acid. Chem Nat Compd 19, 580–582 (1983). https://doi.org/10.1007/BF00576088
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00576088