Abstract
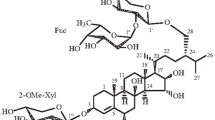
The acid hydrolysis of the total glycosides from the holothurianDuasmodactyla kurilensis Levin. has yielded a new aglycone which has been called kurilogenin. On the basis of the results of NMR, IR, UV, and mass spectroscopy, it has been shown that it is 3β-hydroxy-4,4,14-trimethylpregna-9(11),16-dien-20-one.
Similar content being viewed by others
Literature cited
J. D. Chanley, T. Mezzetti, and H. Sobotka, Tetrahedron,22, 1857 (1966).
B. Tursch, I. S. de Souza Guimaraes, B. Gilbert, R. T. Aplin, A. M. Duffield, and C. Djerassi, Tetrahedron,23, 761 (1967).
W. L. Tan, C. Djerassi, J. Fayos, and J. Clardy, J. Org. Chem.,40, 466 (1975).
G. B. Elyakov, V. A. Stonik, Sh. Sh. Afiyatullov, A. I. Kalinovskii, V. F. Sharypov, and A. Ya. Korotkikh, Dokl. Akad. Nauk SSSR,259, 1367 (1981).
I. Rothberg, B. M. Tursch, and C. Djerassi, J. Org. Chem.,38, 209 (1973).
E. Breitmaier and W. Voelter,13C NMR Spectroscopy, Verlag Chemie, Weinheim-Bergstrasse (1974), p. 210.
N. S. Bhacca, L. F. Johnson, and J. N. Shoolery, NMR Spectra Catalog, No. 352, Instrument Division of Varian Associates, Palo Alto, California (1962).
H. Beierbeck, J. K. Saunders, and J. W. ApSimon, Can. J. Chem.,55, 2813 (1977).
H. Eggert and C. Djerassi, J. Org. Chem.,46, 5399 (1981).
G. B. Elyakov, E. V. Levina, and I. I. Kapustina, Comp. Biochem. Physiol.,55B, 57 (1976).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry, Far Eastern Scientific Center, Academy of Sciences of the USSR, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 6, pp. 724–727, November–December, 1983.
Rights and permissions
About this article
Cite this article
Kalinovskii, A.I., Avilov, S.A., Stepanov, V.R. et al. Glycosides of marine invertebrates. XXIII. Kurilogenin — A new genin from the glycosides of the holothurianDuasmodactyla kurilensis . Chem Nat Compd 19, 688–691 (1983). https://doi.org/10.1007/BF00575170
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00575170