Summary
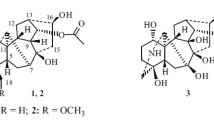
The roots ofAconitum monticola have yielded songorine, songaramine, norsongorine, an amorphous base with the composition C22H35NO6, a base C22H33NO6 with mp 166–167°C, a base C22H33NO5 with mp 161–164°C, and the new alkaloid acomonine. The structure of acomonine has been established on the basis of chemical transformations and spectral properties: It consists of a lycoctonine nucleus with an α-hydroxy group at C3, an α-methoxy group at C10,β-methoxy groups at C6 and C15, an α-glycol system at C7 and C8, and a methoxy group at C19.
Similar content being viewed by others
Literature cited
V. Nezhevenko, M. S. Yunusov, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 409 (1974).
A. S. Narzullaev, M. S. Yunusov, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 497 (1973).
M. S. Yunusov, Ya. V. Rashkes, V. A. Tel'nov, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 515 (1969).
N. Bhacca and D. A. Williams, Applications of NMR Spectroscopy in Organic Chemistry, Holden-Day, San Francisco (1964).
M. S. Yunusov and S. Yu. Yunusov, Khim. Prirodn. Soedin., 85 (1972).
M. S. Yunusov and S. Yu. Yunusov, Khim. Prirodn. Soedin., 335 (1970).
V. A. Tel'nov, M. S. Yunusov, and S. Yu. Yunusov, Khim. Prirodn. Soedin., 583 (1970).
O. Achmatowicz, Jr., Y. Tsuda, and L. Marion, Can. J. Chem.,43, No. 8, 2336 (1965).
Additional information
Institute of Plant Substances, Academy of Sciences of the Uzbek SSR. Translated from Khimiya Prirodnykh Soedinenii, No. 3, pp. 389–395, May–June, 1975.
Rights and permissions
About this article
Cite this article
Nezhevenko, V., Yunusov, M.S. & Yunusov, S.Y. Alkaloids of Aconitum monticola structure of acomonine. Chem Nat Compd 11, 400–404 (1975). https://doi.org/10.1007/BF00571219
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00571219