Conclusions
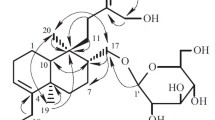
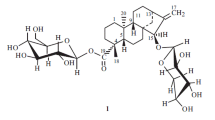
The structure of leontoside E has been established as a hexaoside of hederagenin. Its O-glycosidic moiety is branched and consists of O-β-D-glucopyranosido(1 » 4)-O-β-D-glucopyranosido(1 » 3)-O-α-L-arabopyranose and its O-acyl glycosidic moiety is O-α-L-rhamnopyranosido(1 » 4)-O-β-D-glucopyranosido(1 » 6)-O-β-D-glucopyranose.
Similar content being viewed by others
References
L. G. Mzhel'skaya, V. K. Yatsyn, and N. K. Abubakirov, KhPS [Chemistry of Natural Compounds],2, 421, 1966.
V. G. Bukharov, V. V. Karlin, and V. A. Talan, KhPS [Chemistry of Natural Compounds],3, 17, 1967.
L. G. Mzhel'skaya and N. K. Abubakirov, KhPS [Chemistry of Natural Compounds],3, 101, 218, 1967;4, no. 3, 1968.
J. Polonsky, E. Sach, and E. Lederer, Bull. Soc. Chim. France, 880, 1959.
A. Ya. Khorlin, A. G. Ven'yaminova, and N. K. Kochetkov, Izv. AN SSSR, ser. khim., 1588, 1966.
R. Tschesche, W. Schmidt, and G. Wulff, Z. Naturf.,20b, 708, 1965.
N. K. Kochetkov and A. Ya. Khorlin, DAN SSSR,150, 1289, 1963, N. K. Kochetkov and A. J. Khorlin, Arzneim.-Forsch.,16, 101, 1966.
L. Hough, S. K. N. Jones, and W. H. Wadman, J. Chem. Soc., 1702, 1950.
T. G. Bonner, Chem. Ind., 345, 1960.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 4, No. 4, pp. 216–221, 1968
Rights and permissions
About this article
Cite this article
Mzhel'skaya, L.G., Abubakirov, N.K. Triterpene glycosides ofLeontice eversmannii . Chem Nat Compd 4, 186–189 (1968). https://doi.org/10.1007/BF00571131
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00571131