Conclusions
1. A new base, folifinine, has been isolated from the epigeal part ofHaplophyllum foliosum Vved. Its developed formula is C16H14N(OH)2 (OCH3) (-O-).
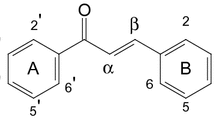
2. The structure 8-hydroxy-7-(3′-hydroxy-3′-methylbutyl)-4-methoxy-2, 3-furoquinoline has been proposed for folifinine.
Similar content being viewed by others
References
Z. Sh. Faizutdinova, I. A. Bessonova, and S. Yu. Yunusov, KhPS [Chemistry of Natural Compounds],3, 257, 1967.
L. H. Briggs and L. D. Colebrook, J. Chem. Soc., 2458, 1960.
Alfred W. Sangster and Kenneth L. Stuart, Chem. Rev.,65, 101, 1965.
R. G. Cooke and H. F. Haynes, Austr. J. Chem.,7, 273, 1954.
N. I. McCorkindale, Tetrah.,14, 223, 1961.
H. Rapoport and K. G. Holden, J. Amer. Chem. Soc.,82, 4395, 1960.
L. M. Jackman, Applications of Nuclear Magnetic Resonance Spectroscopy in Organic Chemistry, Pergamon Press, London,64, 53, 1959.
R. H. Prager, E. Ritchie, A. V. Robertson, and W. C. Taylor, Austr. J. Chem.,15, 301, 1962.
A. V. Robertson, Austr. J. Chem.,16, 451, 1963.
J. A. Diment, E. Ritchie, and W. C. Taylor, Austr. J. Chem.,20, 565, 1967.
K. Nakanishi, IR Spectra and the Structure of Organic Compounds [Russian translation], Moscow, p. 208, 1965.
N. Bhacca and D. Williams, Applications of NMR Spectroscopy in Organic Chemistry [Russian translation], Moscow, p. 129, 1966.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 4, No. 6. pp. 373–376, 1968
Rights and permissions
About this article
Cite this article
Kurbanov, D., Bessonova, I.A. & Yunusov, S.Y. The structure of folifinine. Chem Nat Compd 4, 315–317 (1968). https://doi.org/10.1007/BF00569813
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00569813