Summary
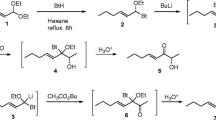
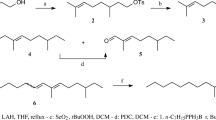
The stereodirected synthesis of 2-methyl-cis-7,8-epoxyoctadecane — the sex attractant ofPorthetria dispar (L.) — has been effected.
Similar content being viewed by others
Literature cited
A. A. Shamshurin, B. G. Kovalev, and A. P. Donya, Dokl. Akad. Nauk SSSR,190, 1362 (1970).
B. A. Bierl, M. Beroza, and C. W. Collier, Science,170, 87 (1970).
E. A. Bried and G. F. Hennion, J. Amer. Chem. Soc.,60, 1717 (1938).
K. Krîz, M. J. Benes, and J. Peska, Tetrahedron Lett., 2881 (1965); Collection Czech. Chem. Commun.,32, 358 (1967).
H. Luftmann and G. Spiteller, Org. Mass-Spectrom.,5, 1073 (1971).
R. T. Aplin and L. Coles, Chem. Commun., 858 (1967).
P. Brown, J. Kossanyi, and C. Djerassi, Tetrahedron Suppl.,8, Part 1, 241 (1966).
K. Eiter, Angew. Chem.,84, 67 (1972).
V. D. Yasnopol'skii, Physicochemical Constants of Organic Compounds with an Acetylenic Bond [in Russian], Baku (1966), p. 15.
I. Heilbron and R. M. Bunbury, Dictionary of Organic Compounds, 2nd ed., Eyre and Spottiswoode, London (1943–4).
Additional information
For Communication I, see [1].
Institute of Chemistry, Academy of Sciences of the Moldavian SSR. Translated from Khimiya Prirodnykh Soedinenii, No. 4, pp. 545–548, July–August, 1973.
Rights and permissions
About this article
Cite this article
Shamshurin, A.A., Rekhter, M.A. & Vlad, L.A. The chemistry of the pheromones II. A new synthesis of disparlure — The sex attractant of the gypsy moth. Chem Nat Compd 9, 509–511 (1973). https://doi.org/10.1007/BF00568640
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00568640