Conclusions
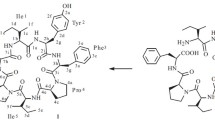
The synthesis of the heptapeptideN ɛ-BOC-N α-pelargonyl-L-lysyl-N ɛ-Z-L-lysyl-D-phenylalanyl-L-leucyl-N ɛ-Z-L-lysyl-N ɛ-Z-L-lysyl-L-threonine, a linear analog of the cyclopeptide part of the antibiotic polymixin B, has been effected (yield 63%). The possibility of using this method for the synthesis of peptides on a polymer support without protecting the hydroxyl group of threonine has been shown.
Similar content being viewed by others
References
W. Hausmann and J. C. Craig, J. Am. Chem. Soc.,76, 4892, 1954.
K. Vogler, R. O. Studer, P. Lanz, W. Lergier, and E. Bohni, Helv. Chim. Acta,48, 1161, 1965.
K. Vogler and R. O. Studer, Exper.,22, 6, 345, 1966.
R. B. Merrifield, J. Am. Chem. Soc.,85, 2149, 1963.
K. Hofmann, R. Schmiechen, R. D. Wells, Y. Wolman, and N. Yanaihara, J. Am. Chem. Soc.,87, 1965.
B. Beras and L. Zervas, J. Am. Chem. Soc.,83, 719, 1961.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 6, No. 3, pp. 359–361, 1970
Rights and permissions
About this article
Cite this article
Morozova, E.A., Zevail', M.A. Synthesis of the heptapeptide of the cyclopeptide moiety of polymixin B. Chem Nat Compd 6, 358–359 (1970). https://doi.org/10.1007/BF00567320
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00567320