Summary
1. A new sesquiterpene lactone which has been called tanacin has been isolated from the leaves ofTanacetum pseudoachillea C. Winkl.
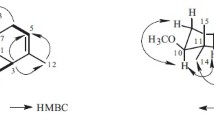
2. On the basis of an interpretation of the PMR spectrum of (I) using the method of multifrequency resonance, the structure of 6-angeloyloxy-1,10-epoxygermacr-4,11(13)-dien-8,12-olide has been established for tanacin.
3. The chemical conversion of tanacin into chrysanin has been performed, which confirms the structure found.
Similar content being viewed by others
Literature cited
A. I. Yunusov, Sh. Z. Kasymov, and G. P. Sidyakin, Khim. Prirodn. Soedin., 276 (1973); 262 (1975).
W. von Philipsborn, Angew. Chem., Int. Ed.,10, 472 (1971).
F. Bohlmann, V. S. Bhaskar Rao, and M. Grenz, Tetrahedron Lett.,36, 3947 (1968).
R. W. Doskotch and F. S. El-Feraly, Can. J. Chem.,47, 1139 (1969).
R. W. Doskotch, F. S. El-Feraly, and C. D. Hufford, Can. J. Chem.,49, 2103 (1971).
M. N. Mukhametzhanov, V. I. Sheichenko, A. I. Ban'kovskii, K. S. Rybalko, and K. I. Boryaev, Khim. Prirodn. Soedin., 56 (1969).
I. S. Akhmedov, Sh. Z. Kasymov, and G. P. Sidyakin, Khim. Prirodn. Soedin., 691 (1970).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 170–174, March–April, 1976.
Rights and permissions
About this article
Cite this article
Yunusov, A.I., Abdullaev, N.D., Kasymov, S.Z. et al. Structure of the sesquiterpene lactone tanacin. Chem Nat Compd 12, 153–156 (1976). https://doi.org/10.1007/BF00566334
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00566334