Summary
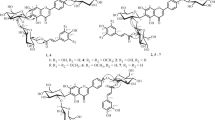
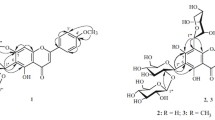
In an investigation of the composition of the extractive substance of the wood ofAbies sibirica Ledeb. andA. nephrolepis Maxim., new O-acylated lignans, previously unknown in nature, have been isolated. The structures of the two new compounds have been established by physicochemical methods and by independent synthesis: they are lariciresinol coumarate and ferulate.
The PMR spectra of laricyl coumarate and laricyl ferulate were taken by G. A. Kalabin.
Similar content being viewed by others
Literature cited
W. J. Taylor and A. R. Battersby, Oxidation Coupling of Phenols, N. J. (1976), p. 276.
S. A. Medvedeva, L. D. Modonova, V. G. Leont'eva, V. N. Glazkova, and N. A. Tyukavkina, Khim. Prirodn. Soedin., 113 (1971).
V. G. Leont'eva, L. D. Modonova, and N. A. Tyukavkina, Khim. Prirodn. Soedin., 268 (1973).
F. Sundom, Acta Chem. Scand.,22, 854 (1973).
L. D. Modonova, V. K. Voronov, V. G. Leont'eva and N. A. Tyukavkina, Khim. Prirodn. Soedin., 165 (1972).
V. G. Leont'eva, L. D. Modonova, and N. A. Tyukavkina, Izv. Sibirskogo Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, No. 4, 159 (1974).
N. S. Vul'fson, Preparative Organic Chemistry [in Russian], Moscow-Leningrad (1964), p. 621.
Additional information
Irkutsk Institute of Organic Chemistry, Siberian Branch of the Academy of Sciences of the USSR. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 162–166, March–April, 1976.
Rights and permissions
About this article
Cite this article
Leont'eva, V.G., Modonova, L.D., Voronov, V.K. et al. New O-acyl derivatives of lariciresinol. Chem Nat Compd 12, 147–150 (1976). https://doi.org/10.1007/BF00566332
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00566332