Abstract
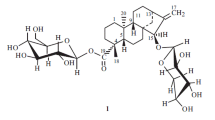
Partial syntheses of glycosides of oleanolic acid — the 3-0-β-D-xylopyranoside (songoroside A), the 3,28-bis-0-β-D-xylopyranoside, and the 28-0-β-gentiobioside-3-0-β-D-xylopyranoside — and also the formation of 3-0-β-D-xylopyranosyloleanolic acid 13,28-lactone are described.
Similar content being viewed by others
Literature cited
A. Akimaliev, P. K. Alimbaeva, L. G. Mzhel'skaya, and N. K. Abubakirov, Khim. Prir. Soedin., 472, 476 (1976).
V. G. Bukharov and V. V. Karlin, Khim. Prir. Soedin., 84 (1969).
S. J. Stolzenberg, R. M. Parkhurst, and E. J. Reist, Contraception,14, 39 (1976).
A. M. Yuodvirshis and A. T. Troshchenko, Izv. Sib. Otd. Akad. Nauk SSSR, Ser. Khim. Nauk, No. 2, 129 (1969).
K. Takamura, Chem. Pharm. Bull.,4, 470 (1956).
A. F. Bochkov and L. G. Kretsu, Izv. Akad. Nauk SSSR, Ser. Khim., 2803 (1971).
E. Hardegger and F. Robinet, Helv. Chem. Acta,33, 1871 (1950);35, 824 (1952).
A. Ya. Khorlin, Yu. S. Ovodov, and N. K. Kochetkov, Zh. Obshch. Khim.,32, 782 (1962).
R. U. Lemieux and J. D. Stevens, Can. J. Chem.,44, 249 (1966).
H. P. Albrecht, Ann. Chem., 1429 (1977).
A. F. Sviridov, L. P. Vecherko, V. I. Kadentsev, O. S. Chizhov, and N. K. Kochetkov, Izv. Akad. Nauk SSSR, Ser. Khim., 2713 (1973).
V. T. Chernobai, Zh. Obshch. Khim.,34, 1018 (1964).
B. Helferich and M. Gindy, Chem. Ber.,87, 1489 (1954).
K. Takiura, S. Honda, T. Endo, and K. Kakehi, Chem. Pharm. Bull.,20, 438 (1972).
J. Becker, Biochim. Biophys. Acta,100, 574 (1965).
Yu. V. Karyakin, Pure Chemical Reagents [in Russian], Moscow-Leningrad (1947), p. 454.
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR, Tashkent. Institute of the Physiology and Experimental Pathology of High Mountain Regions of the Academy of Sciences of the KirgSSR, Frunze. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 668–673, September–October, 1979.
Rights and permissions
About this article
Cite this article
Akimaliev, A.A., Pal'yants, N.S., Alimbaeva, P.K. et al. Triterpene glycosides ofScabiosa soongorica . Chem Nat Compd 15, 587–591 (1979). https://doi.org/10.1007/BF00565931
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00565931