Abstract
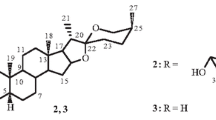
The epigeal part ofArtemisia absinthium L. has yielded a new sesquiterpene lactone artemolin, C15H20O5, with mp 205°C, [α] 20D +18° (c 1.0, chloroform). The structure of artemolin has been established on the basis of chemical transformations and spectral characteristics.
Similar content being viewed by others
Literature cited
F. Bohlmann, P. K. Mahanta, and L. N. Dutta, Phytochemistry,18, 289 (1979).
K. S. Rybalko, Zh. Obshch. Khim.,33, 2734 (1963).
H. Yoshioka, T. Mabry, and B. Timmerman, Sesquiterpene Lactones, University of Tokyo Press (1973), p. 349.
K. Vokač, Z. Samek, V. Herout, and F. Sorm, Collect. Czech. Chem. Commun.,37, 1346 (1972).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 658–661, September–October, 1979.
Rights and permissions
About this article
Cite this article
Kasymov, S.Z., Abdullaev, N.D., Zakirov, S.K. et al. Artemolin — A new guaianolide fromArtemisia absinthium . Chem Nat Compd 15, 577–579 (1979). https://doi.org/10.1007/BF00565928
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00565928