Summary
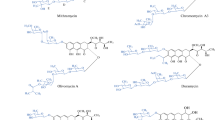
The stereochemical correlation of olivin with chromomycinone and the directed degradation of the first of them to di-O-methyl-D-tartaric acid (XXI) has been effected. As a result, it has been shown that olivin and chromomycinone both have the absolute configuration 2S,3R,1′S,3′S,4′R and possess the respective structures (Ia) and (Ib).
Similar content being viewed by others
Literature cited
Yu. A. Berlin, M. N. Kolosov, and L. A. Piotrovich, Khim. Prirodn. Soedin., 519 (1972).
Yu. A. Berlin, M. N. Kolosov, and L. A. Piotrovich, Tetrahedron Lett., 1329 (1970).
M. Miyamoto, K. Morita, Y. Kawamatsu, K. Kawashima, and K. Nakanishi, Tetrahedron,23, 411 (1967).
M. Miyamoto, K. Morita, Y. Kawamatsu, S. Noguchi, R. Marumoto, M. Sasai, A. Nohara, Y. Nakadaira Y. Y. Lin, and K. Nakanishi, Tetrahedron,22, 2761 (1966).
G. P. Bakhaeva, Yu. A. Berlin, M. M. Kolosov, and O. A. Chuprunova, Khim. Prirodn. Soedin., 580 (1969).
N. Harada, K. Nakanishi, and S. Tatsuoka, J. Amer. Chem. Soc.,91, 5896 (1969).
G. P. Bakhaeva, Yu. A. Berlin, M. N. Kolosov, and O. A. Chuprunova, Khim. Prirodn. Soedin., 572 (1969).
R. T. Aplin and W. T. Pike, Chem. Ind. (London), 2009 (1966); R. W. A. Oliver and R. M. Rashman, J. Chem. Soc., Ser. B, 341 (1971).
R. Anschutz and A. Pictet, Ber.,13, 1176 (1880).
T. Purdie and J. C. Irvine, J. Chem. Soc.,79, 957 (1901).
Additional information
For Communication XXV, see [1], and for a preliminary communication see [2].
M. M. Shemyakin Institute of the Chemistry of Natural Compounds, Academy of Sciences of the USSR. Translated from Khimiya Prirodnykh Soedinenii, No. 4, pp. 526–534, July–August, 1972.
Rights and permissions
About this article
Cite this article
Berlin, Y.A., Kolosov, M.N. & Piotrovich, L.A. Olivomycin and related antibiotics XXVI. The absolute configurations of olivin and of chromomycinone. Chem Nat Compd 8, 515–521 (1972). https://doi.org/10.1007/BF00563684
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00563684