Conclusions
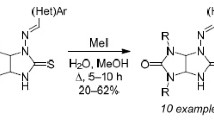
On the basis of information on the kinetics of trans-glycosylation in arylamine N-glycosides, a rapid method has been developed for obtaining urea N-glycosides using m-nitroaniline N-glycosides as donors of glycosyl residues.
Similar content being viewed by others
Literature cited
G. P. Ellis and J. Honeyman, Advan. Carbohydrate Chem.,10, 95 (1955).
V. A. Afanas'ev, F. V. Pishchugin, N. I. Trushkina, and V. A. Kharmats, Homogeneous Catalysis [in Russian], Frunze (1970), p. 41.
V. A. Afanas'ev and F. V. Pishchugin, Zh. Fiz. Khim.,44, 1811 (1970).
F. V. Pishchugin and V. A. Afanas'ev, Zh. Fiz. Khim.,44, 2085 (1970).
A. S. Jones and G. W. Ross, Tetrahedron,18, 189 (1962).
S. A. Barker, E. J. Bourne, R. Stephens, and D. H. Whiffen, J. Chem. Soc., 3468, 4211 (1954).
Additional information
Institute of Organic Chemistry, Academy of Sciences of the Khirgiz SSR. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 176–178, March–April, 1974.
Rights and permissions
About this article
Cite this article
Afanas'ev, V.A., Dzhamanbaev, Z.A. Effective method of obtaining N-glycosides of urea. Chem Nat Compd 10, 187–188 (1974). https://doi.org/10.1007/BF00563610
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00563610