Abstract
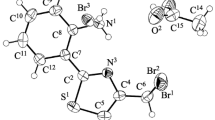
Bromination of the 4-carbethoxy- and 4-carboxy-2-thiazolylhydrazones of aldehydes and ketones occurs principally at the 5 position of the thiazole ring. In the case of aldehyde hydrazones an excess of bromine leads to substitution of the methine hydrogen atom and to intramolecular cyclization of the intermediate halohydrazone to 1,2,4-triazolo[3,4-b]thiazole.
Similar content being viewed by others
References
I. Simiti and M. M. Coman, Bull. Soc. Chim. France, No. 9, 3276 (1969).
A. S. Shawali and C. Parkanyi, J. Heterocycl. Chem., 17, No. 5, 833 (1980).
G. P. Andronnikova, S. L. Nikolaeva, G. S. Balashova, E. I. Boreko, and A. G. Kolomiets, placed in VINITI, No. 139, XII-D83, VINITI Library List, No. 5, 126 (1983).
A. L. Suslova, M. G. Bazhenova, G. I. Validuda, S. L. Nikolaeva, and G. P. Andronnikova, placed in VINITI, No. 825, XII-1084, Russ. Zh. Khim., No. 1, 45 (1985).
R. N. Buttler and F. L. Scott, J. Chem. Soc., C., No, 14, 1711 (1968).
A. A. Tsurkan and A. A. Platonova, Dokl. Akad. Az. SSR, 34, No. 10, 67 (1976).
G. P. Andronnikova, S. V. Usol'tseva, S. L. Nikolaeva, G. M. Anoshina, V. I. Nifontov, and É. M. Emelina, Khim. farm. Zh., No. 11, 1326 (1988).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 257–262, February, 1993.
Rights and permissions
About this article
Cite this article
Usol'tseva, S.V., Andronnikova, G.P. & Shevyrin, V.A. Bromination of 2-thiazolylhydrazones. Chem Heterocycl Compd 29, 226–230 (1993). https://doi.org/10.1007/BF00531672
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00531672