Abstract
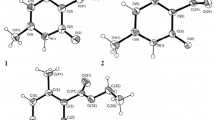
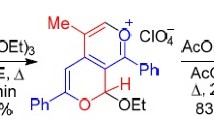
The reaction of 4-phenylethynylpyrilium salts with indoles leads to indolylvinylpyrilium derivatives through attack of the β-carbon atom of the triple bond and subsequent intramolecular transfer of a proton from C(in3) of the indole ring to the sp-hybridized carbon atom of the allene group. The structure of 2,6-diphenyl-4-[2′-phenyl-2′-((β-methylindolyl)vinyl]pyriliumperchloratewasdeterminedunequivocallybyx-ray diffraction analysis and the most likely reaction scheme was indicated. The bond length distribution in the pyrilium cation indicates significant delocalization of positive charge.
Similar content being viewed by others
References
A. V. Koblik, L. A. Murad'yan, A. D. Dubonosov, and G. P. Zolotovskova, Khim. Geterotsikl. Soedin., No. 3, 299 (1990).
A. V. Koblik, L. A. Murad'yan, O. E. Kompan, D. S. Yufit, Yu. T. Struchkov, Yu. A. Zhdanov, L. P. Olekhnovich, and G. S. Zolotovskova, Khim. Geterotsikl. Soedin., No. 7, 885 (1988).
L. A. Murad'yan, G. S. Zolotovskova, and A. V. Koblik, Khim. Geterotsikl. Soedin., No. 3, 307 (1990).
H. Plieninger and D. Wild, Chem. Ber., 99, 3070 (1966).
R. M. Acheson, Khim. Geterotsikl. Soedin., No. 8, 1011 (1976).
H. Berbalk, M. Knollmuller, and C. E. Weikowitsch, Monatsh. Chem., 109, No. 2, 443 (1978).
F. H. Allen, O. Kennard, D. G. Watson, L. Brammer, A. G. Orpen, and R. Taylor, J. Chem. Soc., Perkin Trans. 2, S1 (1991).
L. Pauling, The Nature of the Chemical Bond, Cornell Univ. Press, Ithaca (1960).
T. Tamamura, T. Yamane, N. Yasuoka, and N. Kasai, Bull. Chem. Soc. Jpn., 47, No. 4, 832 (1974).
N. G. Bokii, R. B. Vedrinskii, V. V. Kitaev, N. A. Lopatina, and Yu. T. Struchkov, Koord. Khim., 2, 103 (1976).
O. E. Kompan, K. A. Potekhin, N. G. Furmanova, and Yu. T. Struchkov, Zh. Strukt. Khim. 21, 149 (1980).
L. G. Kuz'mina and Yu. T. Struchkov, Koord. Khim., 9, 705 (1983).
A. V. Goncharov, D. S. Yufit, and Yu. T. Struchkov, Zh. Strukt. Khim., 30, 164 (1989).
D. Chasseau, J. Gaultier, C. Hauw, R. Fugnitto, V. Gianis, and H. Strzelecka, Acta Crystallogr., B38, 1629 (1982).
M. P. Sammes, R. L. Harlow, and S. H. Simonsen, J. Chem. Soc., Perkin Trans. 2, No. 2, 303 (1981).
R. G. Gerr, A. I. Yanovskii, and Yu. T. Struchkov, Kristallografiya, 28, 1029 (1983).
G. N. Dorofeenko, A. V. Koblik, L. A. Murad'yan, T. I. Polyakova, and B. A. Tertov, Zh. Org. Khim., 16, 1741 (1980).
USSR Inventor's Certificate No. 1,189,861; A. V. Koblik, L. A. Murad'yan, S. G. Blagorodov, L. N. Chernavskaya, and N. A. Dmitrieva, Byull. Izobret., No. 41, 103 (1985).
USSR Inventor's Certificate No. 1,244,148; N. A. Dmitrieva, L. N. Chernavskaya, S. G. Blagorodov, A. P. Shepelev, A. V. Koblik, and L. A. Murad'yan, Byull. Izobret., No. 26, 104 (1986).
Author information
Authors and Affiliations
Additional information
For communication 3, see [1]
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 10, pp. 1316–1324, October, 1992.
Rights and permissions
About this article
Cite this article
Koblik, A.V., Murad'yan, L.A., Gridunova, G.V. et al. The ethynyl carbocation. 4. Reaction of 4-phenylethynylpyrylium salts with c-nucleophiles. Chem Heterocycl Compd 28, 1116–1123 (1992). https://doi.org/10.1007/BF00529571
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00529571