Abstract
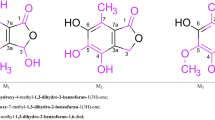
The electrophilic substitution reactions (nitration, bromination, acylation, and the Mannich and Vilsmeier reactions) of 1-benzyl-6-methoxy-7-cyano-5-azaindole and the nitration and Vilsmeier reaction of 6-hydroxy-5-azaindoline were studied.
Similar content being viewed by others
Literature cited
V. A. Azimov, N. N. Bychikhina, and L. N. Yakhontov, Khim. Geterotsikl. Soedin., No. 12, 1648 (1981).
V. A. Azimov, V. G. Granik, R. G. Glushkov, and L. N. Yakhontov, Khim. Geterotsikl. Soedin., No. 3, 355 (1978).
V. A. Azimov, N. N. Bychikhina, and L. N. Yakhontov, Khim. Geterotsikl. Soedin., No. 2, 215 (1981).
L. N. Yakhontov and E. I. Lapan, Khim. Geterotsikl. Soedin., No. 11, 1528 (1972).
S. Okuda and M. M. Robison, J. Org. Chem., 24, 1008 (1959).
A. F. Pozharskii, V. V. Kuz'menko, V. A. Azirnov, and L. N. Yakhontov, Khim. Geterotsikl. Soedin., No. 9, 1232 (1972).
D. M. Krasnokutskaya and L. N. Yakhontov, Khim. Geterotsikl. Soedin., No. 3, 380 (1977).
Author information
Authors and Affiliations
Additional information
See [1] for Communication 60.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 356–360, March, 1982.
Rights and permissions
About this article
Cite this article
Bychikhina, N.N., Azimov, V.A. & Yakhontov, L.N. Azaindole derivatives. 61. Electrophilic substitution reactions in 1-benzyl-6-methoxy-7-cyano-5-azaindole and 6-oxo-5-azaindoline. Chem Heterocycl Compd 18, 268–271 (1982). https://doi.org/10.1007/BF00522126
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00522126