Abstract
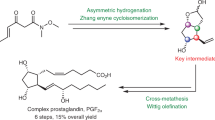
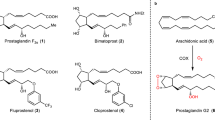
Heterocyclic analogs of prostaglandin, dl-2-(trans-3-hydroxyocten-lyl)-N-(6-ethoxycarbonylhexyl)pyrrole and -indole were obtained by the condensation of 2-formylpyrrole and 2-formylindole with 2-oxoheptylidenetriphenylphosphorane, followed by alkylation with ethyl 7-iodoheptanoate and reduction of the keto group by sodium borohydride.
Similar content being viewed by others
Literature cited
E. I. Levkoeva and L. N. Yakhontov, Usp. Khim., 44, 1074 (1975).
V. A. Dombrovskii, D. Yu. Fonskii, V. A. Mironov, and P. M. Kochergin, Usp. Khim., 53, 689 (1984).
V. G. Avramenko, N. N. Levinova, V. D. Nazina, and N. I. Suvorov, Khim. Geterotsikl. Soedin., No. 2, 204 (1975).
I. Barta, G. Ambrus, Gg. Horvath, M. Sóti, and P. Sóhar, Acta Chim. Sci. Hung., 98, 463 (1978).
G. Ambrus and I. Barta, Prostaglandins, 10, 661 (1975).
P. Parraclough, C. J. Harris, and A. G. Caldwell, U. K. Patent No. 2094793; Ref. Zh. Khim., 12060P (1983).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 40–43, January, 1986.
Rights and permissions
About this article
Cite this article
Dombrovskii, V.A., Gracheva, E.V. & Kochergin, P.M. Synthesis of heterocyclic analogs of prostaglandins from pyrrole and indole. Chem Heterocycl Compd 22, 33–36 (1986). https://doi.org/10.1007/BF00515419
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00515419