Abstract
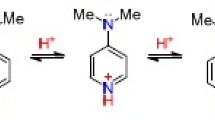
The stepwise protonation of amines can be utilized for the interpretation of the PMR spectra of conjugated pairs consisting of an amine and its salt. A linear dependence of the proton chemical shifts on the composition of the amine-salt system has been demonstrated in the example of 2a-methyl-4-keto-trans-decahydroquinoline, making it possible to determine the proton chemical shifts, which are hidden in one of the partners by other signals, by means of extrapolation. The chemical shifts of the protons nearest the nitrogen atom vary in the following order upon protonation: α-H tertα > α-He terte ≈ β-H tertα > β-H secα ≈ β-H sece ≈ β-Hpr.
Similar content being viewed by others
Literature cited
J. A. Pople, W. G. Schneider, and H. J. Bernstein, High-Resolution Nuclear Magnetic Resonance, McGraw-Hill, New York (1959).
D. V. Sokolov, G. S. Litvinenko, and K. I. Khludneva, Zh. Obshch. Khim., 29, 1112 (1959).
G. S. Litvinenko, A. A. Espenbetov, and Yu. T. Struchkov, Izv. Akad. Nauk Kaz. SSR, Ser. Khim., No. 5, 63 (1982).
A. A. Espenbetov, Yu. T. Struchkov, N. Yu. Kuz'mina, and G. S. Litvinenko, Izv. Akad. Nauk Kaz. SSR, Ser. Khim., No. 5, 59 (1982).
V. F. Bystrov, Usp. Khim., 41, 512 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 11, pp. 1524–1526, November, 1983.
Rights and permissions
About this article
Cite this article
Litvinenko, G.S., Khludneva, K.I. & Krasnomolova, L.P. Amine-ammonium salt systems in the interpretation of PMR spectra. Chem Heterocycl Compd 19, 1210–1212 (1983). https://doi.org/10.1007/BF00515359
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00515359