Abstract
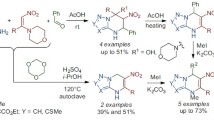
The cycloaddition of 2,5-bis(trifluoromethyl)-1,3,4-oxadiazole to dienophiles with cyclic and acyclic structures takes place according to a two-step mechanism with the extrusion of nitrogen and results in the formation of derivatives of 7-oxabicyclo [2.2.1]heptanes. A significant influence of the donor properties of the dienophiles, as well as of the spatial factors, on the realization of these processes has been discovered. However, solvation efects do not have a significant effect on the formation of the cycloadducts. The regio- and stereoselectivity of the cyclo-addition reactions has been noted.
Similar content being viewed by others
Literature Cited
N. V. Vasil'ev, Yu. E. Kyashenko, A. F. Kolomiets, and G. A. Sokol'skii, Khim. Geterotsikl. Soedin., No. 4, 562 (1987).
M. Palmer, Structure and Reactions of Heterocyclic Compounds, Arnold, London (1967).
D. Berger, Tetrahedron, 39, 2869 (1983).
E. P. Nesynov and A. P. Grekov, Usp. Khim., 33, 1184 (1964).
G. Seitz and H. Wassmuth, Chem. Z., 112, 80 (1988).
F. Thalhammer, V. Wallfahrer, and J. Sauer, Tetrahedron Lett., 29, 3231 (1988).
P. Mhyre, J. Edmonds, and J. Kruger, J. Am. Chem. Soc., 88, 2459 (1966).
H. Van der Plas, Ring Transformation of Heterocycles, Vol. 1, Academic Press, London-New York (1973), pp. 14, 364.
R. Huisgen, R. Grashey, and J. Sauer, in: S. Patai (editor), The Chemistry of Alkenes, Interscience, New York-London (1964) [Russian translation: Khimiya, Moscow (1969), p. 444].
Additional information
For a preliminary report, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 95–100, January, 1990.
Rights and permissions
About this article
Cite this article
Vasil'ev, N.V., Lyashenko, Y.E., Galakhov, M.V. et al. 2,5-bis(trifluoromethyl)-1,3,4-oxadiazole in cycloaddition reactions. Chem Heterocycl Compd 26, 81–85 (1990). https://doi.org/10.1007/BF00506855
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506855