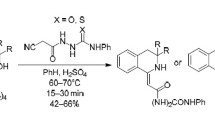
Abstract
It has been shown that α-chloroquinoxalines heteroarylate pyridin-2-yl and quinolin2-ylacetonitriles primarily at the methylene group. A method has been developed for synthesizing 1-R-2-amino-3-heteroarylpyrrolo[2,3-b]quinoxalines permitting the preparation of compounds containing the pyridine and quinoline nuclei.
Similar content being viewed by others
Literature Cited
A. P. Kozynchenko, Yu. M. Volovenko, F. S. Babichev, and V. K. Promonenkov, Ukr. Khim. Zh., 55, 737 (1989).
A. P. Kozynchenko, Yu. M. Volovenko, F. S. Babichev, and V. K. Promonenkov, Khim. Geterotsilk. Soedin., No. 8, 1119 (1988).
F. S. Babichev, Yu. M. Volovenko, A. P. Koznychenko, and V. K. Promonenkov, Russian Patent 1,384,590.
F. S. Babichev, Yu. M. Volovenko, A. G. Nemazanyi, and S. I. Tyukhtenko, Ukr. Khim. Zh., 52, 506 (1986).
K. Winterfield and K. Flick, Arch. Pharm., 289, 448 (1956).
V. Carelli, M. Cardellini, and F. Liberatore, Ann. Chem. (Rome), 49, 709 (1959).
V. D. Romanenko and S. I. Burmistrov, Khim. Geterotsokl. Soedin., No. 6, 852 (1973).
Author information
Authors and Affiliations
Additional information
For Communication 2, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 85–87, January, 1990.
Rights and permissions
About this article
Cite this article
Kozynchenko, A.P., Volovenko, Y.M., Babichev, F.S. et al. Heteroarylation of acetonitriles. 3. Heteroarylation of pyridin-2-yl and quinolin-2-ylacetonitriles by chloroquinoxalines. Chem Heterocycl Compd 26, 73–75 (1990). https://doi.org/10.1007/BF00506853
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506853