Abstract
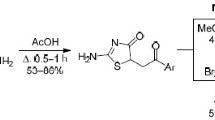
The reaction of arylacetonitriles with methyl thiasalicylate leads to 2-amino-3-aryl-1-thia-4-chromones. The spectral characteristics and the reactions at the amino group were studied.
Similar content being viewed by others
Literature cited
K. Nishibata, M. Nishio, T. Ishii, and S. Ito, Japanese Patent No. 7139865; Chem. Abstr., 76, 46086 (1972).
R. Hazard and J. King, West German Patent No. 2006196; Chem. Abstr., 74, 42279 (1971).
L. A. Zhmurenko, Sh. V. Glozman, and V. A. Zagorevskii, Khim. Geterotsikl. Soedin., No. 2, 182 (1978).
Y. Kawase and K. Sakashita, Bull. Chem. Soc. Jpn., 35, 1869 (1962).
Y. Kawase, Bull. Chem. Soc. Jpn., 32, 9 (1959).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1047–1048, August, 1982.
Rights and permissions
About this article
Cite this article
Volovenko, Y.M., Litenko, V.A., Dashkovskaya, E.V. et al. New synthesis of 2-amino-1-thia-4-chromones. Chem Heterocycl Compd 18, 796–798 (1982). https://doi.org/10.1007/BF00506579
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506579