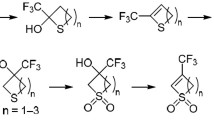
Abstract
Depending on their structure, tetra- and pentasubstituted 4H-thiopyrans either undergo disproportionation or form salts with the participation of air oxygen upon reaction with trifluoroacetic acid. It is shown that these processes are competitive. The previously proposed scheme for the disproportionation of 4H-thiopyrans with protic acids was proved experimentally. The positions at which protonation of the double bonds of the heterorings occurs were established as a result of ionic hydrogenation of the 4H-thiopyrans.
Similar content being viewed by others
Literature cited
V. G. Kharchenko, M. E. Stankevich, N. M. Kupranets, A. R. Yakoreva, V. I. Kleimenova, and S. K. Klimenko, Zh. Org. Khim., 8, 193 (1972).
O. Yu. Okhlobystin and N. T. Berberova, Zh. Org. Khim., 16, 1558 (1958).
I. P. Beletskaya, Zh. Org. Khim., 16, 1560 (1980).
V. G. Kharchenko, N. I. Kozhevnikova, and N. V. Voronina, Khim. Geterotsikl. Soedin., No. 4, 562 (1979).
V. G. Kharchenko and V. I. Kleimenova, Zh. Org. Khim., 7, 613 (1971).
V. G. Kharchenko, V. I. Kleimenova, and A. R. Yakoreva, Khim. Geterotsikl. Soedin., No. 7, 900 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 200–203, February, 1983.
Rights and permissions
About this article
Cite this article
Kharchenko, V.G., Kozhevnikova, N.I. Reaction of polysubstituted 4H-thiopyrans with trifluoroacetic acid in salt-formation and ionic-hydrogenation reactions. Chem Heterocycl Compd 19, 163–166 (1983). https://doi.org/10.1007/BF00506426
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00506426