Abstract
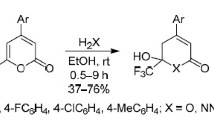
2-Aryl-4-methyl-4,5-epoxytetrahydropyrans, which react with piperidine regioselectively with trans-diaxial cleavage of the oxide ring at the least substituted carbon atom, were synthesized by the reaction of 2-aryl-4-methyl-3,6-dihydropyrans with peracids.
Similar content being viewed by others
Literature cited
A. A. Gevorkyan, A. S. Arakelyan, and N. M. Khizantsyan, Armyansk. Khim. Zh., 30, 743 (1977).
U. G. Ibatullin, D. Ya. Mukhametova, S. A. Vasil'eva, R. F. Talipov, L. V. Syurina, M. G. Safarov, and S. R. Rafikov, Izv. Akad. Nauk SSSR, Ser. Khim., No. 9, 2114 (1982).
V. B. Mochalin, Z. I. Smolina, A. I. Vul'fson, T. I. Dyumaeva, and B. V. Unkovskii, Zh. Org. Khim., No. 7, 825 (1971).
N. S. Vul'fson, G. M. Zolotareva, V. N. Bochkarev, Z. I. Smolina, B. V. Unkovskii, and V. B. Mochalin, Izv. Akad. Nauk USSR, Ser. Khim., No. 2, 437 (1970).
A. A. Gevorkyan, A. S. Arakelyan, L. I. Kazaryan, and G. G. Tokmadzhyan, Arm. Khim. Zh., 30, 685 (1977).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsilklicheskikh Soedinenii, No. 11, pp. 1455–1457, November, 1984.
Rights and permissions
About this article
Cite this article
Ubatullin, U.G., Syurina, L.V., Vasil'eva, S.A. et al. Epoxidation of 2-aryl-4-methyl-3,6-dihydropyrans and reaction of the products with piperidine. Chem Heterocycl Compd 20, 1200–1202 (1984). https://doi.org/10.1007/BF00505705
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00505705