Abstract
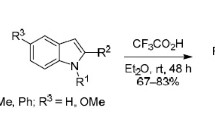
3-Trifluoroacetyl indoles are formed by the action of trifluoroacetic acid on indole-2-carboxylic acid and its benzo-substituted derivatives. When unsubstituted indole is refluxed with trifluoroacetic acid, it gives 3-trifluoroacetylindole in 30% yield.
Similar content being viewed by others
Literature cited
V. A. Budylin, A. N. Kost, and E. D. Matveeva, Khim. Geterotsikl. Soedin., No. 1, 55 (1972).
V. A. Budylin, A. N. Kost, E. D. Matveeva, and V. I. Minkin, Khim. Geterotsikl. Soedin., No. 1, 68 (1972).
V. A. Budylin, E. D. Matveeva, and A. N. Kost, Khim. Geterotsikl. Soedin., No. 9, 1235 (1980).
A. S. Katner, Org. Prep. Proc., 2, 297 (1970).
W. J. Houlighan (editor), Indoles, Part 1, Wiley-Interscience, New York (1972), p. 208.
W. A. Sheppard and C. M. Sharts, The Organic Chemistry of Fluorine [Russian translation], Mir, Moscow (1972), p. 307.
J. Bergman, J. Heterocycl. Chem., 7, 1071 (1970).
Author information
Authors and Affiliations
Additional information
Deceased.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1233–1235, September, 1981.
Rights and permissions
About this article
Cite this article
Kost, A.N., Budylin, V.A., Romanova, N.N. et al. Reaction of indoles with trifluoroacetic acid. Chem Heterocycl Compd 17, 921–923 (1981). https://doi.org/10.1007/BF00505598
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00505598