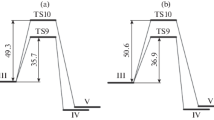
Abstract
The thio-Claisen rearrangement of isomeric 3- and 1-methylallyl phenyl sulfides was investigated. It is demonstrated that the thio-Claisen rearrangement of the 3-methyl isomer is preceded by its thioallyl rearrangement to the 1-methyl isomer. The latter undergoes thio-Claisen rearrangement to o-(3-methylallyl)thiophenol, which is cyclized to 2-ethyl-2,3-dihydrobenzothiophene and 2-methylthiochroman under the reaction conditions.
Similar content being viewed by others
Literature cited
T. A. Danilova, T. Abdin, and E. A. Viktorova, Neftekhimiya, 11, 444 (1971).
T. Abdin, T. A. Danilova, and E. A. Viktorova, Khim. Geterotsikl. Soedin., No. 10, 1337 (1973).
T. Abdin, Master's Dissertation, Moscow State University, Moscow (1974).
H. Kwart and M. H. Cohen, J. Chem. Soc., Chem. Commun., No. 6, 319 (1968).
J. C. Petropoulos, M. A. McCall, and D. S. Tarbell, J. Am. Chem. Soc., 75, 1130 (1953).
H. Kwart and N. A. Johnson, J. Am. Chem. Soc., 92, 6064 (1970).
H. Kwart and N. A. Johnson, J. Am. Chem. Soc., 97, 3441 (1977).
H. Kwart and E. R. Evans, J. Org. Chem., 16, 178 (1958).
H. Kwart and J. L. Schwartz, J. Org. Chem., 39, 1575 (1974).
L. A. Kazitsyna and N. B. Kupletskaya, Application of UV, IR, NMR, and Mass Spectroscopy in Organic Chemistry [in Russian], Izd. MGU, Moscow (1979).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 9, pp. 1191–1194, September, 1981.
Rights and permissions
About this article
Cite this article
Danilova, T.A., Aukharieva, R.G., Petrov, S.N. et al. Mechanism of the thio-claisen rearrangement of 3-methylallyl phenyl sulfide. Chem Heterocycl Compd 17, 883–886 (1981). https://doi.org/10.1007/BF00505590
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00505590