Abstract
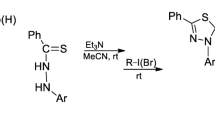
It has previously been observed that the carbamide residue in 2-phenacyl-3-ureido- and 2-cyanomethylpyridine is readily cleaved on cyclization to pyridothiazines [1–4]. It was of interest to establish whether a similar process occurs when the carbonyl component is an α-halo- or unsaturated acid. If this were the case, this reaction could be used to obtain 6-oxopyrido[2,3-b][1,4]thiazines.
Similar content being viewed by others
Literature cited
L. G. Levkovskaya and T. S. Safonova, Khim. Geterotsikl. Soedin., No. 1, 68 (1971).
L. G. Levkovskaya and T. S. Safonova, Khim. Geterotsikl. Soedin., No. 6, 970 (1969).
T. S. Safonova, V. A. Chernov, and N. I. Traven', Inventor's Certificate No. 323405; Byull. Izobret., No. 1 (1972).
N. I. Traven' and T. S. Safonova, Khim. Geterotsikl. Soedin., No. 6, 787 (1980).
T. Takahashi and J. Maki, Pharm. Bull. Jpn., 3, 92 (1955); Chem. Abstr., 50, 10 101 (1956).
L. G. Levkovskaya, N. V. Sazonov, N. A. Grineva, I. E. Mamaeva, L. A. Serochkina, and T. S. Safonova, Khim. Geterotsikl. Soedin., No. 1, 122 (1985).
Author information
Authors and Affiliations
Additional information
For communication 42, see [6].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 214–218, February, 1985.
Rights and permissions
About this article
Cite this article
Traven', N.I., Safonova, T.S. Nitrogen-, oxygen-, and sulfur-containing heterocycles. 43. Synthesis and properties of 6-oxo-5H-pyrido[2,3-b][1,4]thiazines. Chem Heterocycl Compd 21, 176–180 (1985). https://doi.org/10.1007/BF00504204
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00504204