Abstract
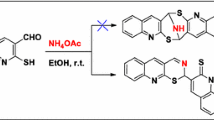
1,4,4a,5,10,10a-Hexahydro-1,3,4-thiadiazino[5,6-b]quinoxalines react with acetylacetone and acetoacetate esters upon heating in ethanol to give derivatives of a novel heterocyclic system, hexahydro-1,3,4-thiadiazolo[2,3-a]quinoxalino[2,3-d]pyrrole.
Similar content being viewed by others
Literature Cited
O. N. Chupakhin, G. M. Petrova, M. G. Ponizovskii, V. N. Charushin, G. G. Aleksandrov, and E. O. Sidarov, Khim. Geterotsikl. Soedin. (1990) (in press).
V. N. Charushin, O. N. Chupakhin, and H. C. van der Plas, Adv. Heterocycl. Chem. 44, 301 (1988).
V. N. Charushin and O. N. Chupakhin, Usp. Khim. 53, 1648 (1984).
N. A. Klyuev, V. G. Baklykov, V. N. Charushin, and O. N. Chupakhin, Khim. Geterotsikl. Soedin., No. 4, 432 (1989).
V. N. Charushin, V. G. Baklykov, O. N. Chupakhin, and V. N. Drozd, Khim. Geterotsikl. Soedin., No. 3, 396 (1985),
V. G. Baklykov, V. N. Charushin, O. N. Chupakhin, and V. N. Drozd, Khim. Geterotsikl. Soedin., No. 4, 557 (1987).
V. N. Charushin, O. N. Chupakhin, and A. I. Rezvukhin, Heterocycles 20, 195 (1981).
V. N. Charushin, V. G. Baklykov, O. N. Chupakhin, and T. Yu. Pushkareva, Khim. Geterotsikl. Soedin., No. 5, 707 (1985).
L. E. Sutton, Tables of Interatomic Distances and Configurations in Molecules and Ions, Supplement 1956–1959, Spec. Publ. Chem. Soc., London (1965), No. 18.
B. Holmberg, Ark. Kemi 9, 65 (1955).
Author information
Authors and Affiliations
Additional information
For Communication 28, see [1].
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 697–702, May, 1990.
Rights and permissions
About this article
Cite this article
Baklykov, V.G., Charushin, V.N., Chupakhin, A.N. et al. Cyclization of N-alkylazinium cations with bifunctional nucleophiles. 29. Synthesis of a novel 1,3,4-thiadiazolo-[2,3-a]quinoxalino[2,3-d]pyrrole heterocyclic system. Chem Heterocycl Compd 26, 592–597 (1990). https://doi.org/10.1007/BF00487440
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00487440