Abstract
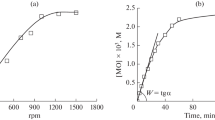
The di-tert-butyl peroxide-initiated (by thermal decomposition) liquid-phase transformations of 2-alkoxytetrahydropyrans to the corresponding esters of n-valeric acid and δ-valerolactone were studied by kinetic methods. Their ratio is determined by the structure of the alkoxy group. The reaction proceeds via a mechanism involving an unbranched chain reaction with quadratic breaking of the chains in the rearranged radicals.
Similar content being viewed by others
Literature cited
D. L. Rakhmankulov, V. S. Martem'yanov, S. S. Zlot-skii, and V. I. Isagulyants, Neftekhmiya, 14, 275 (1974).
É. Kh. Kravets, S. S. Zlot-skii, V. S. Martem'yanov, and D. L. Rakhmankulov, Zh. Prikl. Khim., 48, 2265 (1975).
V. P. Nayanov, D. L. Rakhmankulov, S. S. Zlot-skii, and A. B. Terent'ev, Izv. Akad. Nauk SSSR, Ser. Khim., No. 3, 594 (1975).
C. Berndscom and G. Desctes, Compt. Rend., C280, No. 4, 469 (1975).
E. V. Pastushenko, S. S. Zlot-skii, D. L. Rakhmankulov, and Ya. M. Paushkin, Dokl. Akad. Nauk SSSR, 227, 1409 (1976).
É. Kh. Kravets, Master's Degree, Irkutsk State University (1976).
E. T. Denisov, Rate Constants of Homolytic Liquid-Phase Reactions [in Russian], Nauka, Moscow (1971), p. 258.
L. Fieser and M. Fieser, Reagents for Organic Synthesis [Russian translation], Vol. 1, Mir, Moscow (1971), p. 273.
A. Weissberger, A. Proskauer, and J. Riddick, Organic Solvents [Russian translation], Inostr. Lit., Moscow (1958), p. 83.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh. Soedinenii, No. 4, pp. 456–459, April, 1977.
Rights and permissions
About this article
Cite this article
Pastushenko, E.V., Zlot-skii, S.S. & Rakhmankulov, D.L. Kinetics and mechanism of di-tert-butyl peroxide-initiated liquid-phase transformations of 2-alkoxytetrahydropyrans. Chem Heterocycl Compd 13, 363–366 (1977). https://doi.org/10.1007/BF00482774
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00482774