Abstract
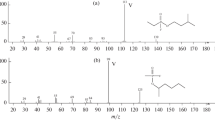
The mass spectra of 2- and 4-iminobarbituric acid derivatives were studied in relation to the mass spectra of their oxygen analogs. It is shown that the pathways of fragmentation of the investigated compounds depend on the type of substituent attached to the C5 atom, the position of the imino and oxo groups in the ring, and the specific mass-spectral properties. The fragmentation was studied by means of low-voltage mass spectrometry and deuterium labeling.
Similar content being viewed by others
Literature cited
V. G. Voronin, S. B. Goncharenko, A. I. Ermakov, and Yu. N. Portnov, Khim. Geterotsikl. Soedin., No. 10, 529 (1976).
M. Mizugaki, Y. Suzuki, M. Uchugama, and H. Abe, Eisei Kagaku, 17, 241 (1971); Chem. Abstr., 76, 68814 (1972).
M. Vore, N. Gerber, and M. T. Bush, Pharmacol., 13, 220 (1971).
I. T. Watson and F. C. Folkner, Mass Spectrom., 7, 1227 (1973).
M. Conrad, Ann., 340, 310 (1905).
M. Conrad and A. Zart, Ann., 340, 326 (1905).
E. Fischer and A. Dilthey, Ann., 335, 352 (1904).
Author information
Authors and Affiliations
Additional information
See [1] for communication I.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 6, pp. 813–817, June, 1978.
Rights and permissions
About this article
Cite this article
Voronin, V.G., Ermakov, A.I., Paramonov, N.K. et al. Mass spectrometry of biologically active substances. Chem Heterocycl Compd 14, 667–671 (1978). https://doi.org/10.1007/BF00481144
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00481144