Abstract
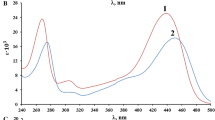
The acid-base properties of 10 derivatives of 2,2′-diquinolyl, as well as quinoline and 5,6-benzoquinoline, were studied by spectrophotometry. It was established that the protonation constants of the systems correlate with the regularities due to the I and M effects of the substituents.
Similar content being viewed by others
Literature cited
N. P. Komar', in: Trudy Nauchno-Issledovat. Instit. Khim. Khar'kovsk. Gosuniversiteta, 8, 61 (1951).
A. L. Gershuns, P. Ya. Pustovar, and V. P. Tkachenko, Vestnik Khar'kovsk. Univ. Khimiya, No 6, 84 (1975).
L. M. Litvinenko, R. S. Popova, and A. F. Popov, Usp. Khim., 14, 1605 (1975).
A. L. Gershuns and P. Ya. Pustovar, Khim. Geterotsikl. Soedin., No. 7 976 (1975).
A. L. Gershuns and P. Ya. Pustovar, Khim. Geterotsikl. Soedin., No. 2, 239 (1973).
A. Weissberger, A. Proskauer, J. Riddick, and A. Toops, Organic Solvents [Russian translation], Inostr. Lit., Moscow (1958).
A. Albert and E. Serjeant, Ionization Constants of Acids and Bases, Methuen (1962).
E. E. Van Hall and K. G. Stone, Anal. Chem., 27, 1580 (1955).
G. A. Laitinen, Chemical Analysis [in Russian], Khimiya, Moscow (1966), p. 580.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 98–100, January, 1977.
Rights and permissions
About this article
Cite this article
Gershuns, A.L., Pustovar, P.Y. Acid-base properties of 2,2′-diquinolyl and its analogs. Chem Heterocycl Compd 13, 84–86 (1977). https://doi.org/10.1007/BF00479876
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00479876