Abstract
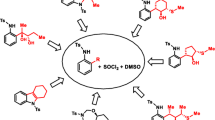
It was established that hard bases replace the methylsulfonyl groups and that soft bases replace the chlorine atoms in 2,4,6-tris(methylsulfonyl)-3,5-dichloropyridine.
Similar content being viewed by others
Literature cited
S. D. Moshchitskii, G. A. Zalesskii, E. A. Romanenko, and V. P. Kukhar', Khim. Geterotsikl. Soedin., No. 3, 369 (1976).
R. G. Pearson, Usp. Khim., 40, 1259 (1971).
S. D. Moshchitskii, G. A. Zalesskii, and L. M. Yagupol'skii, Khim. Geterotsikl. Soedin., No. 6, 802 (1975).
R. G. Pearson and J. Songstad, J. Am. Chem. Soc., 89, 1827 (1967).
H. I. Hertog and I. P. Wibaut, Rec. Trav. Chim., 55, 122 (1936).
J. Bernstein, B. Stearns, M. Dexter, and W. A. Lott, J. Am. Chem. Soc., 69, 1147 (1974).
A. Roedig, K. Grohe, and D. Klatt, Ber., 99, 2818 (1966).
Belgian Patent No. 740278 (1973); Ref. Zh. Khim., 2N164P (1974).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii. No. 7, pp. 915–919, July, 1976.
Rights and permissions
About this article
Cite this article
Moshchitskii, S.D., Zalesskii, G.A. & Kukhar', V.P. Nucleophilic substitution reactions of 2,4,6-tris(methylsulfonyl)-3.5-dichloropyridine. Chem Heterocycl Compd 12, 760–763 (1976). https://doi.org/10.1007/BF00477007
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00477007