Abstract
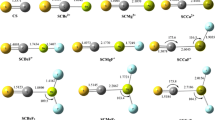
The electronic structure of the thiapyrylium cation with allowance for and neglect of the 3d orbitals of sulfur was investigated by the self-consistent field MO LCAO method within the CNDO/2 (complete neglect of differential overlap) approximation. Inclusion of the 3d AO of sulfur in the basis leads to slight redistribution of the electron density to favor equalization of the charges on the carbon atoms. A qualitative correlation of the calculated populations of the atoms with the chemical shifts of the PMR spectrum of the thiapyrylium cation was obtained.
Similar content being viewed by others
Literature cited
S. Yoneda, T. Sugimoto, and Z. Yoshida, Tetrahedron, 29, 2009 (1973).
G. Karlsson and O. Martensson, Theor. Chim. Acta, 13, 195 (1969).
O. Martensson and C. H. Warren, Acta Chem. Scand., 24, 2745 (1970).
M. E. Dyatkina and N. M. Klimenko, Zh. Strukt. Khim., 14, 173 (1973).
J. P. Pople, D. P. Santry, and G. A. Segal, J. Chem. Phys., 43, Part 2, 129 (1965).
J. A. Pople and G. A. Segal, J. Chem. Phys., 43, Part 2, 136 (1965).
J. A. Pople and G. A. Segal, J. Chem. Phys., 44, 3289 (1966).
J. A. Pople and D. L. Beveridge, Approximate Molecular Orbital Theory, New York (1970), p. 113.
P. A. Howell, R. M. Kurtis, and W. N. Lipscomb, Acta Crystallogr., 7, 498 (1954).
Mazhar-Ul Hague and C. N. Caughlan, J. Org. Chem., 32, 3017 (1967).
E. Boelema, E. J. Visser, and A. Vos, Rec. Trav. Chim., 86, 1275 (1967).
A. Smith, R. Kalish, and E. J. Smutny, Acta Crystallogr., Sect. B, 28, 3494 (1972).
G. D. Andreetti, G. Bocelli, and P. Sgarabotto, J. Chem. Soc., Perkin Trans. II, 1189 (1973).
M. N. Palmer and R. H. Findlay, Tetrahedron Lett., 4165 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1627–1629, December, 1976.
Rights and permissions
About this article
Cite this article
Pronin, A.F., Kharchenko, V.G. & Bagatur'yants, A.A. Role of the 3d orbitals of sulfur in the thiapyrylium cation. Chem Heterocycl Compd 12, 1338–1340 (1976). https://doi.org/10.1007/BF00476721
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476721