Abstract
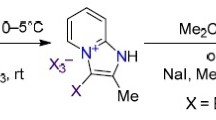
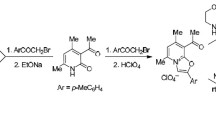
The action of bromine and chlorine of phenacylpyrimidines yields a series of α-mono- and α,α′-dihalophenacylpyrimidines. IR, UV and PMR spectroscopy showed that, in contrast to the starting compounds, the monohalo derivatives exist in the keto form.
Similar content being viewed by others
Literature Cited
Yu. G. Gololobov, P. V. Petrovskii, A. S. Oganesyan, O. A. Zagulyaeva, and V. P. Mamaev, Izv. Akad. Nauk SSSR, Ser. Khim., No. 4, 958 (1984).
O. A. Zagulyaeva and V. P. Mamaev, Izv. Siber. Otdel. Akad. Nauk SSSR, Ser. Khim. Nauk, No. 12, 55 (1967).
S. A. Stekhova, Chemical Sciences Candidate's Dissertation, Novosibirsk (1984).
F. Krönke, Berichte, 69, 516 (1936).
L. Bellamy, The Infrared Spectra of Complex Molecules [Russian translation], Izd. Inos. Lit., Moscow (1963), p. 202.
L. P. Prikazchikova, B. M. Khutova, and E. A. Romanenko, Khim. Geterotsikl. Soedin., No. 9, 1256 (1978).
A. R. Katritsky, H. Z. Kucharska, and J. D. Rowe, J. Chem. Soc., No. 5, 3093 (1965).
R. F. Branch, Nature, 179, 42 (1957).
J. M. Smith, H. W. Steward, B. Roth, and E. H. Northey, J. Am. Chem. Soc., 70, 3997 (1948).
J. Toullec, in: R. W. Taft (editor), Advances in Physical Organic Chemistry, Vol. 18, Academic Press, London (1982), p. 1.
Z. Budesinsky, Coll. Czech. Chem. Commun., 14, 223 (1949).
V. A. Dyatlov, Yu. G. Gololobov, P. V. Petrovskii, A. A. Espenbekov, Yu. T. Struchkov, O. A. Zagulyaeva, and V. P. Mamaev, Izv. Akad. Nauk SSSR, Ser. Khim., No. 11, 2497 (1986).
Author information
Authors and Affiliations
Additional information
Deceased.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 5, pp. 663–667, May, 1987.
Rights and permissions
About this article
Cite this article
Semushkina, I.V., Zagulyaeva, O.A. & Mamaev, V.P. Halogenation of phenacylpyrimidines. Chem Heterocycl Compd 23, 550–553 (1987). https://doi.org/10.1007/BF00476386
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476386