Abstract
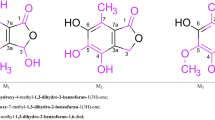
The structures of p-hydroxystyrylpyridines and a number of their new quaternary salts were studied by means of electronic absorption spectra. The reverse transformation of the salts to the corresponding quinonoid compounds was examined as a function of the pH of the medium, and the equilibrium constants were determined.
Similar content being viewed by others
Literature cited
G. N. Bogdanov, A. N. Rozenberg, and A. K. Sheinkman, Khim. Geterotsikl. Soedin., 1660 (1971).
V. V. Ershov and G. A. Nikiforov, Usp. Khim., 35, 1953 (1966).
A. R. Katritzky and J. M, Lagowski, in: Advances in Heterocyclic Chemistry, Vol. 1, New York- London (1963), p. 347.
A. K. Sheinkman, A. N. Kost, V. I. Sheichenko, and A. N. Rozenberg, Ukr. Khim. Zh., 33, 941 (1967).
V. V. Ershov and G. A. Nikiforov, Izv. Akad. Nauk SSSR, Ser. Khim., 293 (1964).
Z. Hortwitz, J. Org. Chem., 21, 1039 (1956).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1087–1091, August, 1972.
Rights and permissions
About this article
Cite this article
Rozenberg, A.N., Bogdanov, G.N. & Sheinkman, A.K. Structures of sterically hindered p-hydroxystyrylpyridines and acid-base transformations of their quaternary salts. Chem Heterocycl Compd 8, 982–985 (1972). https://doi.org/10.1007/BF00476328
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00476328